PTSD BIOMARKERS
"In 1980, the American Psychiatric Association (APA) added PTSD to
the third edition of its Diagnostic and Statistical Manual of Mental
Disorders (DSM-III) nosologic classification scheme. Although
controversial when first introduced, the PTSD diagnosis has filled an
important gap in psychiatric theory and practice.
https://www.ptsd.va.gov/professional/treat/essentials/history_ptsd.asp#:~:text=In%201980%2C%20the%20American%20Psychiatric,in%20psychiatric%20theory%20and%20practice
[2132]
Accounts of PTSD-like symptoms can be found in the Epic of Gilgamesh,
the writings of Herodotus and Hippocrates, and in the Indian epic poem
Ramayana.
Terms used of the years included shell shock, soldier's heart,
irritable heart, Da Costa's syndrome, combat fatigue or war neurosis.
And for a a couple of centuries "nostalgia" - not its present
meaning:
"In the late 1600s, Swiss physician Dr. Johannes Hofer coined the
term 'nostalgia' to describe Swiss soldiers who suffered from despair
and homesickness, as well as classic PTSD symptoms like sleeplessness
and anxiety. Around the same time, German, French and Spanish doctors
described similar illnesses in their military patients.
"In 1761, Austrian physician Josef Leopold Auenbrugger wrote about
nostalgia in trauma-stricken soldiers in his book Inventum Novum. The
soldiers, he reported, became listless and solitary, among other
things, and efforts could do little to help them out of their
torpor.
"PTSD in the Civil War
Nostalgia was a phenomenon noted throughout Europe and the 'disease'
reached American soil during the U.S. Civil War (1861-1865). In fact,
nostalgia became a common medical diagnosis that spread throughout
camps. But some military doctors viewed the illness as a sign of
weakness and one that only affected men with a 'feeble will' - and
public ridicule was sometimes the recommended 'cure' for
nostalgia."
https://www.history.com/topics/inventions/history-of-ptsd-and-shell-shock
[2133]
Shivitti is a sort of case study, The author was sufficiently
impressed by his psychedelic outcome that he wrote a book about
it:
"An autobiographical record of one Holocaust survivor's triumph over
concentration camp syndrome with a regimen of professionally
administered LSD.
"Imprisoned in Auschwitz for two years, having eluded death by the
narrowest of margins, the man known as Ka-tzetnik 135633 survived the
Holocaust to discover that survival alone would not end his torment.
For over 30 years, through nightly dreams of terrifying intensity, the
writer remained captive to the horrors of Auschwitz. Finally in 1976
he sought help from Professor Jan Bastiaans, the Dutch psychiatrist
who first recognized Concentration Camp Syndrome and successfully
treated camp survivors with a therapy involving doses of LSD. Shivitti
is a memoir of that experience."
https://maps.org/product/shivitti-a-vision/
[3548]
Trauma author Dr David Healy charts the course of its treatment from
the Civil War, before and after Freudianism, to the post-Vietnam
intervention of the DSM and its gradual generalisation to to the
traumas common in life, to today. Slovenia's own slant on all of this
is an uncooked omlette.
"The word shellshock essentially rebranded hysteria, which up till WW
I had been a disorder of women and people with weaker constitutions. A
new name was needed because in WW I officers, no less than the working
class men in trenches, were badly affected. Women in contrast were
becoming surgeons and running hospitals back at home. The social order
was shaken up. There was an uptick of births linked to WW I but not as
dramatic as the increase 30 years later.
"Shellshock helped make Freud's reputation. Paradoxically, Freud came
to the fore not because he believed trauma was a factor causing
nervous problems but because his position was that trauma, whether War
or Sexual Abuse in childhood, was irrelevant. People broke down in War
or in Life, because they had pre-existing adjustment difficulties
linked to poor prior management of their libidinous impulses. Current
difficulties (trauma) played into the prior weakness. See Shipwreck in
Maastricht.
"This view excuses the powers that be from causing our nervous
breakdowns. It stops us suing governments for drafting us into
military or vaccine wars. It lays a basis for pointing to a
pre-existing mendacity of women when they claim sexual abuse or
rape.
"Thinking on these lines was not unique to Freud. It played a growing
part in industrial accidents and 'mental trauma' linked to these
accidents in American legal cases from the creation of railroads in
the 1850s onwards.
"It shaped how the American military dealt with the question of
military pensions after the Civil War - ultimately creating in the US
what might be called the first single party payer health service
anywhere in the world. It colored how Germany and Britain dealt with
Shellshock after WW I.
"For WW II the Americans screened men for psychological weakness and
excluded them. This did nothing to eliminate the problems. The War and
the post War nervous problems entrenched psychoanalytic thinking in
the United States. The Europeans went to War armed with high dose
barbiturates, which helped in many but not all cases.
"Analysis came out of the War strengthened. This led to claims that
it was obvious where the War had come from - the Germans were
maladjusted. Democrat voting psychiatrists, unlike Republican voting
doctors in general, knew what was wrong with Republican politicians
like Goldwater and Reagan - they were latent homosexuals. In an era
when Americans put a man on the moon, the shrinks figured it was
within reach to prevent Wars by curing our neuroses.
"Data on personality profiles showing that German concentration camp
guards were more normal and less psychopathic than the American G.I.s
who liberated the camps did nothing to change minds on this."
...and his analysis comes to a frightening conclusion about
industrialized medicine.
Today we seek biological explanations - to produce targeted cures.
But PTSD biomarkers remain a topic of some complexity with little
opportunity for access to analyses for ordinary patients.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4520791/
[51]
List of potential tests in Table 1
The last biomarker mentioned is lowered endocannabinoids, and
reference 64 refers to
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3870889/
[52]
in which AEA and 2-AG were assayed in addition to cortisol in
survivors of the WTC collapse, so the patients were all exposed to
similar experiences at the same time, increasing its usefulness
"The effect of reduced 2-AG content in PTSD remained significant
after controlling for the stress of exposure to the WTC collapse,
gender, depression and alcohol abuse. There were no significant group
differences for AEA or cortisol levels; however, across the whole
sample AEA levels positively correlated with circulating cortisol, and
AEA levels exhibited a negative relationship with the degree of
intrusive symptoms within the PTSD sample. This report shows that PTSD
is associated with a reduction in circulating levels of the eCB 2-AG.
Given the role of 2-AG in the regulation of the stress response, these
data support the hypothesis that deficient eCB signaling may be a
component of the glucocorticoid dysregulation associated with PTSD.
The negative association between AEA levels and intrusive symptoms is
consistent with animal data indicating that reductions in AEA promote
retention of aversive emotional memories."
The ventromedial prefrontal cortex may contribute to conditioned
response inhibition via suppression of the amygdala.
https://www.jneurosci.org/content/jneuro/26/37/9503.full.pdf
[148]
Hwang et al (2025) relate how...
"Recent studies have begun to detail the molecular biology of PTSD.
However, given the array of PTSD-perturbed molecular pathways
identified so far, it is implausible that a single cell type is
responsible. Here we profile the molecular responses in over two
million nuclei from the dorsolateral prefrontal cortex of 111 human
brains, collected post-mortem from individuals with and without PTSD
and major depressive disorder. We identify neuronal and non-neuronal
cell-type clusters, gene expression changes and transcriptional
regulators, and map the epigenomic regulome of PTSD in a
cell-type-specific manner. Our analysis revealed PTSD-associated gene
alterations in inhibitory neurons, endothelial cells and microglia and
uncovered genes and pathways associated with glucocorticoid
signalling, GABAergic transmission and neuroinflammation."
https://www.nature.com/articles/s41586-025-09083-y
[5255]
The locus coeruleus (LC) is a small, bluish nucleus located in the
brainstem, specifically in the pons. It is situated in the floor of
the fourth ventricle, just under the cerebellum. This tiny structure
serves as the brain's primary source of norepinephrine, a
neurotransmitter crucial for functions such as attention, arousal,
sleep-wake cycles, and memory.
Engborg et al (2025) found "Stress reactivity is modulated by
cannabinoid type-1 receptors in norepinephrine and epinephrine neurons
in a context-dependent manner":
"Here, we identify a role for CB1R in NE/E neurons in regulating
stress-related affective responses using a combination of
intersectional genetic, anatomical, behavioral, and physiological
approaches. In control mice, Cnr1 mRNA was widely expressed in
medullary C1/A1 and C2/A2 nuclei, with more limited expression in LC
neurons, highlighting molecular diversity within the central
noradrenergic system. Notably, Cnr1 was present in both NE- and
E-producing medullary neurons, marking the first cell-type-specific
characterization of Cnr1 in brainstem NE/E populations and extending
prior work focused primarily on the LC (Luskin et al., 2024, Oropeza
et al., 2007, Srivastava et al., 2022, Wyrofsky et al., 2017). In
Cnr1cKO-Dbh mice, Cnr1 expression was reduced across all examined NE/E
nuclei. Behaviorally, Cnr1cKO-Dbh mice exhibited increased center time
following restraint stress, reduced immobility in the forced swim
test, heightened active escape responses to a looming visual threat,
and diminished rearing and ambulation during the initial threat
presentation. In contrast, no genotype differences were observed in
the elevated zero maze or light-dark box following foot shock stress,
nor in the tail suspension test. Additionally, heart rate dynamics
remained unchanged. These findings suggest that CB1R signaling in NE/E
neurons modulates behavioral responses to acute stress in a
context-dependent manner."
https://www.sciencedirect.com/science/article/pii/S0306452225008942
[5367]
In "Childhood Trauma is Associated with Poorer Cognitive Performance
in Older Adults" (2018) Petkus et al say:
"Neurobiological mechanisms involving chronic inflammation, decreased
neuroplasticity, and epigenetic modification of stress-related
pathways may explain the possible association between early life
trauma and poorer cognitive performance in later life. The
hypothalamus-pituitary-adrenal (HPA) axis is thought to play a
particularly important role in this association. The HPA axis
activates under stress, resulting in elevated levels of the cortisol
hormone and certain severe and/or chronic stressors, particularly if
experienced early in life, may permanently alter HPA axis function.
Studying this association is important, as chronically elevated
cortisol levels have been associated with worse neuropsychological
performance in later life."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6959209/
[3583]
"Perceived stress can have long-term physiological and psychological
consequences
and has shown to be a modifiable risk factor for Alzheimer disease
and related dementias." say Kulshreshtha et al (2023), who compared
perceived stress and health indicators:
"The final analytical sample included 24 448 participants (14 646
women [59.9%]; median age, 64 years [range, 45-98 years]; 10 177 Black
participants [41.6%] and 14 271 White participants [58.4%]). A total
of 5589 participants (22.9%) reported elevated levels of stress.
Elevated levels of perceived stress (dichotomized as low stress vs
elevated stress) were associated with 1.37 times higher odds of poor
cognition after adjustment for sociodemographic variables,
cardiovascular risk factors, and depression (adjusted odds ratio
[AOR], 1.37; 95% CI, 1.22-1.53). The association of the change in the
Perceived Stress Scale score with incident cognitive impairment was
significant in both the unadjusted model (OR, 1.62; 95% CI, 1.46-1.80)
and after adjustment for sociodemographic variables, cardiovascular
risk factors, and depression (AOR, 1.39; 95% CI, 1.22-1.58). There was
no interaction with age, race, and sex."
https://doi.org/10.1001/jamanetworkopen.2023.1860
[3584]
The state of knowledge on species differences shows that humanity is
at an early stage in the understanding of these processes.
https://www.pnas.org/content/pnas/early/2019/12/17/1902288116.full.pdf
[149]
But according to Ney et al at the School of Psychology, University of
Tasmania, much of the research as it relates to PTSD and cannabinoids
may be gender-skewed:
"Despite men reportedly experiencing more traumatic events than
women, lifetime prevalence of PTSD is twice as common in women in
Australia (Silove et al., 2017), United Kingdom (Wittchen et al.,
2011) and the United States (Kessler et al., 1995; Tolin Foa, 2006).
Whilst exposure to different trauma types (and specifically
interpersonal and sexual violence in women) makes a substantial
contribution, this is insufficient to explain the difference in
prevalence between the sexes (Olff, Langeland, Draijer, & Gersons,
2007; Tolin & Foa, 2006). Despite sex hormones being effectors of
stress responses critical to PTSD aetiology, the majority of
psychological and pharmacological PTSD preclinical research is
conducted using males (Lebron-Milad & Milad, 2012). It is
therefore possible that treatments developed for PTSD may be
insensitive to the specific needs of female patients"
See their Table 3 for an overview of studies on sex differences and
endocannabinoid signalling,
https://www.researchgate.net/profile/Luke-Ney-2/publication/326337009_Modulation_of_the_endocannabinoid_system_by_sex_hormones_Implications_for_Posttraumatic_Stress_Disorder/links/5fbd8bfda6fdcc6cc663e25d/Modulation-of-the-endocannabinoid-system-by-sex-hormones-Implications-for-Posttraumatic-Stress-Disorder.pdf
[4387]
Maldonado et al in "The endocannabinoid system in modulating fear,
anxiety, and stress" (2020) explain more about stress and the
ECS:

"Stress is an alteration of homeostasis as a consequence of external
or internal threats. Indeed, acute stressors elicit immediate and
protracted neuroendocrine responses with protective effects. These
responses involve the activation of the sympathetic nervous system and
the HPA axis. Within seconds of stress exposure, noradrenaline and
adrenaline are released through sympathetic postganglionic neurons and
adrenal gland chromaffin cells contributing to fight-or-escape
protective responses. In parallel, the HPA axis is activated through
CRH release from the hypothalamus, which leads to ACTH release from
the pituitary gland. ACTH in the general blood stream reaches adrenal
glands that pour glucocorticoids into the blood circulatory system.
These corticoids activate glucocorticoid receptors that increase
glucose availability and trigger transcriptional changes partly
directed to limit inflammation and repair processes that can be
postponed.
"The endocannabinoid system present in the HPA axis and the
sympathetic nervous system plays a crucial role in regulating stress
responses (Figure 3). Early studies showed that repeated THC modified
dopamine b-hydroxylase activity in rodent serum, a measure of
sympathetic system activation. This effect was different depending on
the basal status of exposed subjects revealing a complex modulatory
role of the endocannabinoid system: THC alleviated sympathetic
activation in naive mice but potentiated this response in rodents
subjected to immobilization stress. CB1R controls peripheral and
central adrenaline, and noradrenaline release involved in
stress-induced memory impairment, and the sympathetic nervous system
partly mediates the anxiety-like effects observed after CB1R blockade.
In regard to the HPA axis, pharmacological and knockout studies
demonstrate that CB1R activity limits hypothalamic CRH release. CB1R
is also present in the pituitary gland and adrenal cortex cells, where
it restricts ACTH and glucocorticoid release, respectively. On the
contrary, glucocorticoids induce fast increases in endocannabinoid
synthesis in brain areas that shape the perception of psychological
stressors. These regions include areas involved in cognitive processes
such as PFC and hippocampus, and areas related with affective
responses such as the amygdala. Glucocorticoids released after acute
stressors activate G-protein membrane receptors in the BLA, promoting
a rapid increase in retrograde 2-AG signaling that leads to
suppression of GABAergic synaptic inputs onto BLA principal neurons,
inducing fast increases in anxiety-like behavior. These limbic areas
are further connected with the hypothalamus to modulate stress
responses (Figure 3). CB1R located in these structures represent a
unique opportunity for pharmacological modulation. However, the use of
exogenous CB1R ligands has been associated with serious health
problems, from THC effects promoting addictive behaviors and anxiety
disorders to the psychiatric alterations (anxiety, depression, or
suicidal behaviors) related to the CB1R inverse agonist rimonabant.
Therefore, a great deal of research has focused on the pharmacological
modulation of endogenous cannabinoids as an alternative approach for
the treatment of stress-induced alterations. In this context, the
enzymes involved in the synthesis and degradation of endocannabinoids
represent potential effective targets with possible better safety
profiles than drugs directly acting on CB1R."
And
"Local CB1R activity has also been associated with anxiogenesis in
certain circuits. Thus, the amygdala sends CB1R -positive projections
to the bed nucleus of the stria terminalis (BNST), a brain region
closely involved in anxiety. This area is connected with midbrain
structures, including the ventral tegmental area and the locus
coeruleus. Glutamatergic and GABAergic projections from the amygdala
to BNST are each sufficient for the development of anxious responses
to unpredictable stimuli. CB1R activity in these projections seems
essential for the shift from phasic to sustained fear responses in
fear conditioning chambers, interpreted as a reminiscence of the human
anxiety against unpredictable threats. In the context of an
established anxiogenic response, blocking cannabinoid activity in the
BNST facilitates the transition from persistent to phasic freezing
responses, a switch that could allow earlier reinstatement of normal
anxious behavior."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7605023/
[2942]
New data keeps pouring in on THC and it all tends to support the
notion that THC is a fear-reducing substance. More precisely, one
which inhibits the system by which learned fear is retained. Reporting
the "Influence of Δ9-tetrahydrocannabinol on long-term neural
correlates of threat extinction memory retention in humans" (2019)
Hammoud et al say:
"...our results are the first to examine the long-term impact of a
single dose administration of THC on the functional activation of the
threat extinction network. Our findings show a significant effect on
the functional connectivity of threat-detection network that emerged
after a week from engagement. These data highlight the need to further
investigate the long-term influence of THC on threat and anxiety
circuitry. Specifically, THC, or compounds with comparable impact on
CB1 receptors (e.g., cannabidiol) could be used as adjuncts to
extinction-based therapies for PTSD and anxiety disorders. This is
especially relevant to PTSD treatment given that threat extinction
learning and extinction memory retention has been shown to be
deficient in PTSD patients. Moreover, the neural correlates related to
PTSD psychopathology are comparable to those engaged by THC in the
present study."
https://www.nature.com/articles/s41386-019-0416-6.pdf
[150]
"The Neuropeptide Y (NPY)-ergic System is Associated with Behavioral
Resilience to Stress Exposure in an Animal Model of Post-Traumatic
Stress Disorder" reported Cohen et al, who frightened some mice with a
predator scent before testing, in 2011.
"Animals whose behavior was extremely disrupted (EBR) selectively
displayed significant downregulation of NPY in the hippocampus,
periaqueductal gray, and amygdala, compared with animals whose
behavior was minimally (MBR) or partially (PBR) disrupted, and with
unexposed controls. One-hour post-exposure treatment with NPY
significantly reduced prevalence rates of EBR and reduced trauma-cue
freezing responses, compared with vehicle controls. The distinctive
pattern of NPY downregulation that correlated with EBR as well as the
resounding behavioral effects of pharmacological manipulation of NPY
indicates an intimate association between NPY and behavioral responses
to stress, and potentially between molecular and psychopathological
processes, which underlie the observed changes in behavior. The
protective qualities attributed to NPY are supported by the extreme
reduction of its expression in animals severely affected by the
stressor and imply a role in promoting resilience and/or recovery."
https://pmc.ncbi.nlm.nih.gov/articles/PMC3242318/
[6057]
In 2017 Sandberg et al showed "Low neuropeptide Y in cerebrospinal
fluid in bipolar patients is associated with previous and prospective
suicide attempts", noting that
"No biological markers that help predict suicide been
identified....Patients were reexamined one year after the lumbar
puncture and suicide attempts were recorded. NPY-LI was significantly
lower in patients with a history of suicide attempt than in patients
who had not attempted suicide prior to lumbar puncture. Importantly,
NPY-LI was markedly lower in patients who made a suicide attempt
during the follow-up period compared to patients who did not. Patients
who attempted suicide during the follow-up also had markedly lower
NPY-LI than those with previous suicide attempts who did not
reattempt. Our results suggest that low CSF NPY-LI predicts future
suicide attempts."
https://www.sciencedirect.com/science/article/abs/pii/S0924977X14002855?via%3Dihub
[6058]
Among a series of studies of fear extinction involving Irit Akirev
[3987], is "Neuropeptide Y and cannabinoids interaction in the
amygdala after exposure to shock and reminders model of PTSD" from
Mayman et al (2020):
"Modulation of cannabinoid and neuropeptide Y (NPY) receptors may
offer therapeutic benefits for post-traumatic stress disorder (PTSD).
In this study, we aimed to investigate the functional interaction
between these systems in the basolateral amygdala (BLA) in a rat model
of PTSD.
"Rats were exposed to the shock and reminders model of PTSD and
tested for hyper arousal/PTSD- and depression-like behaviors 3 weeks
later. Immediately after shock exposure rats were microinjected into
the BLA with URB597, a selective inhibitor of fatty acid amide
hydrolase (FAAH) that increases the levels of the endocannabinoid
anandamide or with the NPY1 receptor agonist Leu31,Pro34-NPY
(Leu).
"Intra-BLA URB597 prevented the shock/reminders-induced PTSD-
behaviors (extinction, startle) and depression-behaviors (despair,
social impairments). These preventing effects of URB597 on PTSD- and
depression-like behaviors were shown to be mostly mediated by
cannabinoid CB1 and NPY1 receptors, as they were blocked when URB597
was co-administered with a low dose of a CB1 or NPY1 receptor
antagonist. Similarly, intra-BLA Leu prevented development of all the
behaviors. Interestingly, a CB1 antagonist prevented the effects of
Leu on despair and social behavior, but not the effects on extinction
and startle. Moreover, exposure to shock and reminders upregulated CB1
and NPY1 receptors in the BLA and infralimbic prefrontal cortex and
this upregulation was restored to normal with intra-BLA URB597 or
Leu.
"The findings suggest that the functional interaction between the eCB
and NPY1 systems is complex and provide a rationale for exploring
novel therapeutic strategies that target the cannabinoid and NPY
systems for stress-related diseases."
https://www.sciencedirect.com/science/article/abs/pii/S0028390819303661
[3989]
In 2025, 100 years since General Smuts got his way at the Opium
Convention, Xue et al were happy to reveal "An Amygdala-hippocampus
Circuit for Endocannabinoid Modulation of Anxiety Avoidance":
"Recent studies indicate a therapeutic potential of increased brain
endocannabinoids (eCBs) in anxiety disorders, but the underlying brain
circuits are still elusive. Here, it is observed that optogenetic
inhibition and activation of anterior basolateral amygdala (aBLA) -
ventral hippocampus (vHPC) glutamatergic projections respectively
decrease and increase anxiety avoidance behaviors. Then, the
contributions of eCBs in aBLA-vHPC projections to anxiety avoidance
are investigated by employing three newly developed synapse- and
circuit-specific eCB-targeted viral strategies to achieve real-time
monitoring of eCB release, in vivo optogenetic activation of CB1
receptors, and CRISPR-Cas9 gene knockdown of eCB biosynthesis enzymes.
Prominent eCB release are surprisingly found at aBLA-vHPC
glutamatergic synapses during anxiety avoidance, suggesting inhibitory
effects of increased eCBs in aBLA-vHPC projections on anxiety
avoidance. This idea is further supported by findings that specific
activation of CB1 receptors at aBLA-vHPC synapses inhibit presynaptic
glutamate release and reduce anxiety avoidance. In contrast, specific
knockdown of eCB biosynthesis enzymes at aBLA-vHPC synapses reduce eCB
levels at aBLA-vHPC glutamatergic synapses and increase anxiety
avoidance. Additionally, inhibition of aBLA-innervated vHPC
glutamatergic neurons alleviates anxiety avoidance. Together, these
findings reveal counteracting effects of increased eCB signaling in
aBLA-vHPC circuits on anxiety avoidance."
https://advanced.onlinelibrary.wiley.com/doi/pdfdirect/10.1002/advs.202505121
[5521]
Nobuo Masataka (2025) frightened some naive cats with fake
thunderstorms. CBD helped:
"In humans, cannabidiol (CBD), the primary non-addictive component of
cannabis, is known to possess considerable therapeutic potential. The
purpose of this study was to investigate the effects of CBD
administration on reducing sound-induced fear in healthy domestic cats
in a laboratory model of thunderstorm simulation. A total of 40 cats,
each naive to the current testing, were randomly assigned into either
of two administration groups (CBD and placebo). Each group was then
exposed to the thunderstorm test twice; once at the beginning of the
administration (the administration of CBD at 4.0 mg/kg/day over a
2-week-period or the administration of the same amount of sunflower
oil as a placebo) and once after the end of the administration. When
undesirable urination was observed, occurrences of this behaviour were
found to decrease significantly when CBD was administered. However, no
such changes were recorded when the placebo was administered. These
results indicate that CBD could be an effective option for the
treatment of noise-induced fear."
https://www.mdpi.com/2076-2615/15/11/1642
[5341]
For "Cannabidiol Treatment in a Predator-Based Animal Model of PTSD:
Assessing Oxidative Stress and Memory Performance" Jîtcă et al
(2025) found:
"CBD exhibited a tendency to reduce anxiety, a common symptom of
PTSD, although this effect was not statistically significant. However,
it demonstrated protective effects on memory, as evidenced by the MWM
test. Likewise, CBD also showed a reduction in MDA levels, which
implies that an improvement in PTSD symptomatology may also target a
reduction in oxidative stress. Since there are several models used to
induce PTSD, we cannot consider that one is better than the other,
because each brings an advantage, which allows us to create a picture
of the mechanisms involved in the occurrence of PTSD and how the
symptoms can be improved. An important direction of this topic could
be the investigation of the molecular mechanisms by which CBD
influences PTSD symptoms, including the analysis of the expression of
genes and proteins involved in the stress response, neuroinflammation
and neuroplasticity (BDNF) and inflammatory markers (IL-6,
TNF-α) in order to provide additional information about the mode
of action of CBD."
https://www.mdpi.com/1422-0067/26/10/4491/pdf?version=1746705743
[5034]
Then there was Sultan et al (2025) "Controlled Inhalation of
Tetrahydrocannabinol-Predominant Cannabis Flos Mitigates Severity of
Post-Traumatic Stress Disorder Symptoms and Improves Quality of Sleep
and General Mood in Cannabis-Experienced UK Civilians: A Real-World,
Observational Study". Beginning with the observation that
"Approximately 4% of the UK population experiences PTSD", this study
of 58 victims is outlined in Figure 1, showing improvements in global
PTSD score, intrusion, avoidance, cognition and mood, and
hyperreactivity/hyperarousal:

https://karger.com/mca/article-pdf/7/1/149/4282163/000540978.pdf
[5106]
"Endocannabinoids inhibit contextual fear memory generalization via
hippocampal GABAergic synaptic transmission" report Ge et al
(2025):
"Memory generalization allows an organism to adapt to new conditions,
but overgeneralization of fear or traumatic experiences can be
detrimental to survival and contributes to the development of various
mental disorders. However, the cellular and molecular mechanisms
underlying fear memory generalization, especially in the hippocampus,
remain largely unknown. In this study, utilizing a well-established
mouse model of fear memory generalization, we investigated the role of
endocannabinoids (eCBs)-mediated GABAergic synaptic inputs to
hippocampal pyramidal neurons in regulating contextual fear memory
generalization. Our results revealed that pharmacological or genetic
blockade of CB1R in hippocampal CA1 resulted in overgeneralization of
contextual fear memory but not fear memory expression. Subsequent
investigations in conditional knockout mice revealed the involvement
of CB1R in GABAergic neurons, but not those in glutamatergic neurons
or astrocytes, in this overgeneralization. In addition, activation of
GABAA receptors on pyramidal neurons was required for inducing
overgeneralization via AM281, a CB1R antagonist. Neural mechanistic
studies showed that eCBs/CB1R signaling regulates both the activity
and plasticity of inhibitory synapses during generalization,
highlighting the prominence of the disinhibition of CB1R in
interneurons during this process. Subsequently, we delved into the
downstream effects and found that eCB-dependent long-term potentiation
(LTP) in CA1 pyramidal neurons was regulated by the aforementioned
mechanisms. Our findings illustrate that the eCBs/CB1R signaling
pathway modulates the balance between fear memory discrimination and
generalization by controlling inhibitory inputs to hippocampal
pyramidal neurons, accompanied by alterations in excitatory plasticity
within this region."
https://www.pnas.org/doi/10.1073/pnas.2423974122
[5254]
"Medical Cannabis Improved PTSD Symptoms, Anxiety, and Quality of Life
Over an 18-Month Period" say Datta et al (2025):
"In 269 patients, significant improvements in PTSD symptoms, anxiety,
sleep quality, and HRQoL were observed at all follow-up points
(p < 0.001). On multivariate logistic regression,
male gender (OR = 0.51; 95% CI:0.28-0.94;
p = 0.034) was associated with a reduced chance of
reporting improvements in IES-R. Adverse events were reported by 70
(26.02%) patients, with insomnia (n = 42, 15.61%) and
fatigue (n = 40, 14.87%) being the most common."
Nevertheless sleep quality scale (SQS) improved significantly, along
with nearly all other parameters: avoidance, intrusions, hyperarousal,
self-care, usual activities, pain and discomfort, anxiety and
depression, generalized anxiety disorder‐7 (GAD-7), patient global
impression of change (PGIC) and impact of event scale revised (IES-R)
total score at all follow-up points: 1, 3, 6, 12 and 18 months.

https://www.tandfonline.com/doi/full/10.1080/14737175.2025.2490539#d1e649
[5179]
Diurnal variations in endocannabinoids
https://pubmed.ncbi.nlm.nih.gov/31610409/
[53]
Shows daily variation in endocannabinoids, the effect of circadian
rhythms, on AEA, 2-AG and congeners OEA and PEA.
"Congeners of AEA, the structural analogs oleoylethanolamide (OEA)
and palmitoylethanolamide (PEA) were simultaneously assayed. Prior to
24-h blood sampling, each participant was exposed to two nights of
normal (8.5 h) or restricted sleep (4.5 h). The two sleep conditions
were separated by at least one month. In both sleep conditions, during
the period of blood sampling, each individual ate the same
high-carbohydrate meal at 0900, 1400, and 1900.
"Results: Mean 24-h concentrations of AEA were 0.697 ± 0.11
pmol/ml. A reproducible biphasic 24-h profile of AEA was observed with
a first peak occurring during early sleep (0200) and a second peak in
the mid-afternoon (1500) while a nadir was detected in the mid-morning
(1000). The 24-h profiles for both OEA and PEA followed a similar
pattern to that observed for AEA. AEA, OEA, and PEA levels were not
affected by sleep restriction at any time of day, contrasting with the
elevation of early afternoon levels previously observed for
2-AG.
"Conclusions: The 24-h rhythm of AEA is markedly different from that
of 2-AG, being of lesser amplitude and biphasic, rather than
monophasic. These observations suggest distinct regulatory pathways of
the two eCB and indicate that time of day needs to be carefully
controlled in studies attempting to delineate their relative roles.
Moreover, unlike 2-AG, AEA is not altered by sleep restriction,
suggesting that physiological perturbations may affect AEA and 2-AG
differently. Similar 24-h profiles were observed for OEA and PEA
following normal and restricted sleep, further corroborating the
validity of the wave-shape and lack of response to sleep loss observed
for the AEA profile. Therapeutic approaches involving agonism or
antagonism of peripheral eCB signaling will likely need to be tailored
according to time of day."
These two papers tell us that a) reduced EC is a reliable marker of
PTSD; b) more AEA correlates with less symptoms of PTSD; c) AEA
behaves differently in its diurnal behaviour; and d) any perceived
need for exogenous supplementation of EC is likely to be time of day
dependent.
"In a study of the effects of THC on fear extinction, Compared to
subjects who received placebo, participants who received THC showed
increased vmPFC and HIPP activation to a previously extinguished
conditioned stimulus (CS+E) during extinction memory recall."
https://pmc.ncbi.nlm.nih.gov/articles/PMC7001881/
[146]
Another study using MRI and a well-established threat-processing
paradigm found that low doses of ∆9-tetrahydrocannabinol (THC) can
produce anxiolytic effects, reduce threat-related amygdala activation,
and enhance functional coupling between the amygdala and medial
prefrontal cortex and adjacent rostral cingulate cortex (mPFC/rACC)
during threat processing in healthy adults.
https://pubmed.ncbi.nlm.nih.gov/32162103/
[147]
Recognising the cannabis (but not its illegality) sometimes produces
anxiety, in 2015 Komaki et al of the Neurophysiology Research Center,
Hamadan University of Medical Sciences, Iran, evaluated anxiety and
CB1 effects:
"Previous studies have shown that [the] cannabinoidergic system is
involved in anxiety. However, there are controversial reports in the
experimental studies. The aim of this study is to evaluate the effect
of pharmacological stimulation or blocking of CB1 receptors and
inhibition of endocannabinoid degradation in anxiety like behavior in
elevated plus-maze (EPM) test in rat. The EPM is one of the most
widely used animal models of anxiety.
"Male Wistar rats were randomly allocated to ten groups. Different
groups of animals intraperitoneally received Win-55212 (0.3, 1 and 5
mg/kg) as CB1 receptor agonist, AM-251 (0.3, 1 and 5 mg/kg) as CB1
receptor antagonist, URB-597 (0.03, 0.1 and 0.3 mg/kg) as
endocannabinoid breakdown inhibitor or saline (as control group) 30
min before submitting into EPM test.
"The results showed that compared to the control group, Win-55212 (1
and 5 mg/kg) and URB-597 (0.1 and 0.3 mg/kg) significantly increased
both of the time and percentage of entries into open arms. AM-251 (1
and 5 mg/kg) significantly decreased the time and percentage of
entries into open arms in the EPM test. These substances have no
effects on the total distance covered by animals and number of closed
arm entries.
"It is concluded that activation of cannabinoid receptor exert
anxiolytic effect while blocking of cannabinoid receptor resulted in
anxiety behavior. The locomotor activity was not significantly changed
by cannabinoid system. It is suggested that potentiation of
cannabinoid system may be therapeutic strategy for the anxiety
behavior."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4656987/
[2108]
In 2010 El-Alfy et al at the University of Mississippi had tried some
different anxiety tests:
"The antidepressant action of cannabis as well as the interaction
between antidepressants and the endocannabinoid system has been
reported. This study was conducted to assess the antidepressant-like
activity of Δ9-THC and other cannabinoids. Cannabinoids were
initially evaluated in the mouse tetrad assay to determine doses that
do not induce hypothermia or catalepsy [a neurological finding of
prolonged muscular rigidity and immobility with decreased response to
external stimuli and reduced sensitivity to pain]. The automated mouse
forced swim (FST) and tail suspension (TST) tests were used to
determine antidepressant action. At doses lacking hypothermic and
cataleptic effects (1.25, 2.5, and 5 mg/kg, i.p.), both Δ9-THC
and Δ8-THC showed a U-shaped dose response with only
Δ9-THC showing significant antidepressant-like effects at 2.5
mg/kg (p < 0.05) in the FST. The cannabinoids cannabigerol (CBG)
and cannabinol (CBN) did not produce antidepressant-like actions up to
80 mg/kg in the mouse FST, while cannabichromene (CBC) and cannabidiol
(CBD) exhibited significant effect at 20 and 200 mg/kg, respectively
(p < 0.01). The antidepressant-like action of Δ9-THC and CBC
was further confirmed in the TST. Δ9 -THC exhibited the same
U-shaped dose response with significant antidepressant-like action at
2.5 mg/kg (p < 0.05) while CBC resulted in a significant dose
dependent decrease in immobility at 40 and 80 mg/kg doses (p <
0.01). Results of this study show that Δ9-THC and other
cannabinoids exert antidepressant-like actions, and thus may
contribute to the overall mood-elevating properties of
cannabis."
https://europepmc.org/backend/ptpmcrender.fcgi?accid=PMC2866040&blobtype=pdf
[2109]
Akirav (2013) summarised the resilience-promoting properties of
cannabinoids thus:
"Glucocorticoid and endocannabinoid systems cross-talk after
stress.
Exogenous cannabinoid agonists decrease HPA axis activity after
stress.
Stress and glucocorticoids modulate endocannabinoid levels in limbic
areas.
Cannabinoid receptor agonists ameliorate the effects of stress on
emotional memory.
Cannabinoids decrease HPA axis activity in the amygdala via the
GABAergic system."
She proposed the involvement of the amygdala:
"Enhancing cannabinoids signaling using exogenous CB1 receptor
agonists prevent the effects of acute stress on emotional memory. I
propose a model suggesting that the ameliorating effects of
exogenously administered cannabinoids on emotional learning after
acute stress are mediated by the decrease in the activity of the HPA
axis via GABAergic mechanisms in the amygdala."
... which we shall see was later confirmed below.
https://www.sciencedirect.com/science/article/abs/pii/S0149763413001929?via%3Dihub
[3986]
On "Neuroplasticity in Posttraumatic Stress Disorder"
López-López and Crespo (2025) (machine translated from
Spanish) tell us:
"Resilience, understood as the dynamic adaptation following trauma,
is a determining factor in the vulnerability to develop PTSD that may
be associated with myelination. It has been described that contextual
fear memory, like other memories, requires the generation of new
myelin; therefore, the use of promyelinating drugs such as clemastine
fumarate in mice improves remote memory recall and favors fear
generalization. In war veterans with and without PTSD, estimating the
degree of myelination through T1/T2-weighted magnetic resonance
imaging, a positive correlation has been observed between the total
index of the Clinician-Administered PTSD Scale (CAPS) and myelination
in the hippocampus. Furthermore, this same correlation is observed
regarding the severity of depressive symptoms.
"A recent translational study focusing on the hippocampus, amygdala
and corpus callosum described similar results in rats and humans. In
rodents, analysis of myelination in the gray matter of the hippocampal
dentate gyrus shows a positive correlation between oligodendrocyte
density and myelin basic protein, a protein that is translated only in
mature myelinating oligodendrocytes, and the avoidance and anxiety
phenotype, while in the amygdala and cornu ammonis (CA) regions of the
hippocampus this positive correlation occurs with contextual fear
learning. This corroborates data from a previous study using this same
protein, together with specific oligodendrocytes of the hippocampal
dentate gyrus, showing that they are associated with hypervigilance,
avoidance and escape in the face of stress from two weeks after its
onset."
https://pmc-ncbi-nlm-nih-gov.translate.goog/articles/PMC12326437/?_x_tr_sl=auto&_x_tr_tl=en&_x_tr_hl=sl
[5246]
Zhu et al (2025) add that:
"The bed nucleus of stria terminalis (BNST) acts as a crucial hub for
assessing vigilant threats, with the oval subnucleus (ovBNST) being
enriched in endocannabinoid ligands and receptors. The endocannabinoid
system (ECS) is well recognized for its role in stress responses.
However, the molecular and circuitry mechanisms through which the
ovBNST ECS mediates chronic stress induced depressive phenotypes
remain unclear.
"Methods and results: The chronic unpredictable mild stress (CUMS)
was optimized to model the depression-like behaviors and body weight
loss in mice. By utilizing the endocannabinoid sensor, an increased
release of endocannabinoid in the ovBNST was probed in response to
acute stress. Local blockage of ovBNST cannabinoid type 1 receptor
(CB1R) with NESS0327 induced both anhedonia and despair depressive
phenotypes in naive mice. In contrast, intra-ovBNST infusion of either
CB1R agonist or cannabinoid hydrolase inhibitor JZL-184 ameliorated
despair-like behaviors while merely changed anhedonia in CUMS mice. By
combining viral tracing with RNAscope and western blotting, the
reduction in CB1R transcriptional and translational level was found to
be associated with the CUMS induced depressive disorders. This
reduction may be attributed to the changes in ovBNST located
presynaptic CB1R that originates from the medial prefrontal cortex
(mPFC).
"Discussion: Overall, these results suggest that chronic stress may
restructure the ovBNST ECS to result in depressive phenotypes. This
study may extend the comprehension of ECS in the ovBNST, specifically
its role in modulating the pathogenesis of depressive disorders
induced by chronic stress."
https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1629351/full
[5271]
According to Forsythe and Boileau in "Use of cannabinoids for the
treatment of patients with post-traumatic stress disorder"
(2021):
"Numerous treatment options have been developed for the disorder,
including psychotherapeutic approaches such as exposure therapy,
virtual reality therapy, cognitive behavioral therapies and eye
movement desensitization and reprocessing. Around 40-70% of
individuals receiving these treatments briefly have noticed
significant improvement in symptoms and extinction of the learned
fear. Not all individuals are ideal candidates for these treatments,
however. High rates of suicidality, dissociation, destructive
impulsivity, and chaotic life problems are indications that the
patients may not respond well to treatment, causing clinicians to
become more likely to forgo attempting these therapies. This exposes a
need for developing other therapy options."
https://www.degruyter.com/document/doi/10.1515/jbcpp-2020-0279/html
[3546]
"SSRIs have long been considered first-line pharmacological treatment
for PTSD; however, only about 60% of patients respond to it with only
20-30% reaching full remission. The most promising agents to aid in
the process of improving PTSD symptoms were found to target receptors
such as the N-methyl-D-aspartic acid (NMDA) receptor, the receptors
targeted by 3,4-methylenedioxyN-methylamphetamine (MDMA), such as the
5HT2 receptor, and the endocannabinoid CB1 receptor."
Ten studies THC, CBD or synthetic cannabinoids are summarised in
Table 1.
"A common, and expected, limitation of these studies is small sample
size."
https://www.degruyter.com/document/doi/10.1515/jbcpp-2020-0279/pdf
[297]
As Rabinak et al (2020) reported in "Cannabinoid modulation of
corticolimbic activation to threat in trauma-exposed adults: a
preliminary study":
"In adults with PTSD, THC lowered threat-related amygdala reactivity,
increased mPFC activation during threat, and increased mPFC-amygdala
functional coupling." said Wayne State University Detroit researchers
Rabinak et al in "Cannabinoid modulation of corticolimbic activation
to threat in trauma-exposed adults: a preliminary study" (2020).
"During fMRI scanning, participants completed an emotional face
processing task developed by Hariri and colleagues (Hariri et al.
2002) that has been shown to reliably elicit threat-related amygdala
responses."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7244361/
[4558]
Earlier in 2013 Rabinak et al had considered the "Cannabinoid
facilitation of fear extinction memory recall in humans":
"We conducted a study using a randomized, double-blind,
placebo-controlled, between-subjects design, coupling a standard
Pavlovian fear extinction paradigm and simultaneous skin conductance
response (SCR) recording with an acute pharmacological challenge with
oral dronabinol (synthetic THC) or placebo (PBO) 2 h prior to
extinction learning in 29 healthy adult volunteers (THC = 14; PBO =
15) and tested extinction retention 24 h after extinction learning.
Compared to subjects that received PBO, subjects that received THC
showed low SCR to a previously extinguished CS when extinction memory
recall was tested 24 h after extinction learning, suggesting that THC
prevented the recovery of fear. These results provide the first
evidence that pharmacological enhancement of extinction learning is
feasible in humans using cannabinoid system modulators, which may thus
warrant further development and clinical testing.
"This article is part of a Special Issue entitled 'Cognitive
Enhancers'
"Highlights
► Δ9-tetrahydrocannabinol (THC) facilitates extinction of
conditioned fear in humans. ► THC attenuated spontaneous recovery of
fear (vs. placebo). ► THC did not affect within-session extinction
learning. ► Cannabinoids may facilitate exposure-based therapy in
patients with anxiety."
https://www.sciencedirect.com/science/article/abs/pii/S0028390812003371
[6023]
In 2019, Lake et al of the British Columbia Centre on Substance Use
asked "Does cannabis use modify the effect of post-traumatic stress
disorder on severe depression and suicidal ideation? Evidence from a
population-based cross-sectional study of Canadians". And the answer
was yes.
"Among 24,089 eligible respondents, 420 (1.7%) reported a current
clinical diagnosis of post-traumatic stress disorder. In total, 106
(28.2%) people with post-traumatic stress disorder reported past-year
cannabis use, compared to 11.2% of those without post-traumatic stress
disorder (p < 0.001). In multivariable analyses, post-traumatic
stress disorder was significantly associated with recent major
depressive episode (adjusted odds ratio = 7.18, 95% confidence
interval: 4.32-11.91) and suicidal ideation (adjusted odds ratio =
4.76, 95% confidence interval: 2.39-9.47) among cannabis non-users.
post-traumatic stress disorder was not associated with either outcome
among cannabis-using respondents (both p > 0.05)."
https://journals.sagepub.com/doi/abs/10.1177/0269881119882806
[4560]
Nacasch et al (2022) targeted the endocannabinoid system in
treatment-resistant combat PTSD:
"In this retrospective naturalistic study, we followed 14 relatively
mature (32-68 years of age), treatment-resistant, chronic combat
post-traumatic patients who remained severely symptomatic despite
treatment with many lines of conventional treatment prior to receiving
medicinal cannabis. Our findings show that total sleep score,
subjective sleep quality, and sleep duration significantly improved (p
< 0.01). Total PTSD symptom score and its subdomains
(intrusiveness, avoidance, and alertness) showed improvement (p <
0.05). However, there was no improvement in the frequency of
nightmares (p = 0.27). The mean follow-up time was 1.1 ± 0.8
years (range of 0.5 to 3 years)."
https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2022.1014630/full
[4559]
In "Self-reported Medicinal Cannabis Use as an Alternative to
Prescription and Over-the-counter Medication Use Among US Military
Veterans" (2023) McNabb et al examined a mostly RML-friendly
Massachusetts-based cohort of ex-armed forces personnel, reporting in
Clinical Therapeutics.
"A total of 510 veterans of US military service participated in the
survey, which was administered between March 3 and December 31, 2019.
The participants reported experiencing a variety of mental and other
physical health conditions. Primary health conditions reported
included chronic pain (196; 38%), PTSD (131; 26%), anxiety (47; 9%),
and depression (26; 5%). Most participants (343; 67%) reported using
cannabis daily. Many reported using cannabis to reduce the use of
over-the-counter medications (151; 30%) including antidepressants
(130; 25%), anti-inflammatories (89; 17%), and other prescription
medications. Additionally, 463 veterans (91% of respondents) reported
that medical cannabis helped them to experience a greater quality of
life and 105 (21%) reported using fewer opioids as a result of their
medical cannabis use. Veterans who were Black, who were female, who
served in active combat, and who were living with chronic pain were
more likely to report a desire to reduce the number of prescription
medications they were taking (odds ratios = 2.92, 2.29, 1.79, and
2.30, respectively). Women and individuals who used cannabis daily
were more likely to report active use of cannabis to reduce
prescription medication use (odds ratios = 3.05 and 2.26)."
https://www.clinicaltherapeutics.com/article/S0149-2918(23)00133-9/fulltext
[2859]
Another survey of 52 veterans, all with chronic pain, found that
cannabis was the most frequently used natural products, followed by
magnesium and turmeric (or curcumin) and vitamins.

https://escholarship.org/content/qt4896q9pd/qt4896q9pd_noSplash_62d21e2a7124ce26aee1df003cbcfb0f.pdf?t=ssqyzq
[4841]
In the USA Bonn-Miller et al (2022)
"...assessed PTSD symptoms and functioning every 3 months over the
course of a year in two samples of participants diagnosed with PTSD:
(1) those with PTSD using dispensary-obtained cannabis (cannabis
users) and (2) those with PTSD, who do not use cannabis (controls).
Linear mixed-effects models and generalized estimating equations
tested whether trajectories of symptoms differed between the two
subsamples.
"Results: A total of 150 participants (mean [standard deviation] age,
50.67 [15.26] years; 73% male) were enrolled in the study. Over the
course of 1 year, the cannabis users reported a greater decrease in
PTSD symptom severity over time compared to controls [group×time
interaction=−0.32 (95% confidence interval [CI]=−0.59 to
−0.05, R2=0.13; t=−2.35, p=0.02). Participants who used
cannabis were 2.57 times more likely to no longer meet DSM-5 criteria
for PTSD at the end of the study observation period compared to
participants who did not use cannabis (95% CI=1.12-6.07;
p=0.03)."
and
"Adjusting for age and veteran status, cannabis users showed a
significantly greater rate of decline for hyperarousal symptoms
compared to controls."

https://www.ptsd.va.gov/professional/articles/article-pdf/id1563675.pdf
[2104]
In Israel, Nasach et al examined "Medical cannabis for
treatment-resistant combat PTSD" (2023).
"In this retrospective naturalistic study, we followed 14 relatively
mature (32-68 years of age), treatment-resistant, chronic combat
post-traumatic patients who remained severely symptomatic despite
treatment with many lines of conventional treatment prior to receiving
medicinal cannabis. Our findings show that total sleep score,
subjective sleep quality, and sleep duration significantly improved (p
< 0.01). Total PTSD symptom score and its subdomains
(intrusiveness, avoidance, and alertness) showed improvement (p <
0.05). However, there was no improvement in the frequency of
nightmares (p = 0.27). The mean follow-up time was 1.1 ± 0.8
years (range of 0.5 to 3 years)."

https://www.frontiersin.org/articles/10.3389/fpsyt.2022.1014630
[2359]
McNabb et al examined "Self-reported Medicinal Cannabis Use as an
Alternative to Prescription and Over-the-counter Medication Use Among
US Military Veterans" (2023):
"A total of 510 veterans of US military service participated in the
survey, which was administered between March 3 and December 31, 2019.
The participants reported experiencing a variety of mental and other
physical health conditions. Primary health conditions reported
included chronic pain (196; 38%), PTSD (131; 26%), anxiety (47; 9%),
and depression (26; 5%). Most participants (343; 67%) reported using
cannabis daily. Many reported using cannabis to reduce the use of
over-the-counter medications (151; 30%) including antidepressants
(130; 25%), anti-inflammatories (89; 17%), and other prescription
medications. Additionally, 463 veterans (91% of respondents) reported
that medical cannabis helped them to experience a greater quality of
life and 105 (21%) reported using fewer opioids as a result of their
medical cannabis use. Veterans who were Black, who were female, who
served in active combat, and who were living with chronic pain were
more likely to report a desire to reduce the number of prescription
medications they were taking (odds ratios = 2.92, 2.29, 1.79, and
2.30, respectively). Women and individuals who used cannabis daily
were more likely to report active use of cannabis to reduce
prescription medication use (odds ratios = 3.05 and 2.26)."
and
In line with previous research and VA statistics, the participants in
this study experienced several mental and other physical health
concerns. Many reported experiences with PTSD, exposure to
occupational environmental hazards (eg, agent orange, asbestos, burn
pits), chronic pain, anxiety, and depression, among other conditions.
Study findings also revealed that participants' cannabis use
positively affected the treatment or management of several health
conditions and symptoms. Many of the respondents reported that
medicinal cannabis treatment helped them to experience a greater
quality of life, fewer psychological symptoms, fewer physical
symptoms, and to use less alcohol, fewer medications, less tobacco,
and fewer opioids.
https://www.clinicaltherapeutics.com/article/S0149-2918(23)00133-9/fulltext#seccesectitle0001
[2823]
According to Lynskey et al (2024):
"Background
Cannabis-based medicinal products (CBMPs) are increasingly being used
to treat post-traumatic stress disorder (PTSD), despite limited
evidence of their efficacy. PTSD is often comorbid with major
depression, and little is known about whether comorbid depression
alters the effectiveness of CBMPs.
"Aims
To document the prevalence of depression among individuals seeking
CBMPs to treat PTSD and to examine whether the effectiveness of CBMPs
varies by depression status.
"Method
Data were available for 238 people with PTSD seeking CBMP treatment
(5.9% of the treatment-seeking sample) and 3-month follow-up data were
available for 116 of these. Self-reported PTSD symptoms were assessed
at treatment entry and at 3-month follow-up using the PTSD Checklist -
Civilian Version (PCL-C). The probable presence of comorbid depression
at treatment entry was assessed using the nine-item Patient Health
Questionnaire (PHQ-9). Additional data included sociodemographic
characteristics and self-reported quality of life.
"Results
In total, 77% met screening criteria for depression, which was
associated with higher levels of PTSD symptomatology (mean 67.8 v.
48.4, F(1,236) = 118.5, P < 0.001) and poorer general health,
quality of life and sleep. PTSD symptomatology reduced substantially 3
months after commencing treatment (mean 58.0 v. 47.0, F(1,112) = 14.5,
P < 0.001), with a significant interaction (F(1,112) = 6.2, P <
0.05) indicating greater improvement in those with depression (mean
difference 15.3) than in those without (mean difference 7).
"Conclusions
Depression is common among individuals seeking CBMPs to treat PTSD
and is associated with greater symptom severity and poorer quality of
life. Effectiveness of CBMPs for treating PTSD does not appear to be
impaired in people with comorbid depression."
https://www.cambridge.org/core/journals/bjpsych-open/article/medicinal-cannabis-for-treating-posttraumatic-stress-disorder-and-comorbid-depression-realworld-evidence/F88B15F3E00576E757ADF4915F9874A6
[4483]
In a larger study "Suicidal Ideation in Medicinal Cannabis Patients:
A 12-Month Prospective Study" (2024) the objective of Lynskey et al
was...
"Objective
To document the prevalence and correlates of suicidal ideation (SI)
among individuals seeking cannabis-based medicinal products (CBMPs);
to test whether SI declines or intensifies after three months of CBMP
treatment and to document 12-month trajectories of depression in those
reporting SI and other patients.
"Method
Observational data were available for 3781 patients at entry to
treatment, 2112 at three months and 777 for 12 months.
Self-reported depressed mood and SI were assessed using items from the
PHQ-9. Additional data included sociodemographic characteristics and
self-reported well-being.
"Results
25% of the sample reported SI at treatment entry and those with SI
had higher levels of depressed mood (mean = 17.4 vs. 11.3; F(1,3533) =
716.5, p < .001) and disturbed sleep (mean = 13.8 vs. 12.2,
F(1,3533) = 125.9, p < .001), poorer general health (mean = 43.6
vs. 52.2, F(1,3533) = 118.3, p < .001) and lower quality of life
(mean = 0.44 vs. 0.56 (F(1,3533) = 118.3, p < .001). The prevalence
of SI reduced from 23.6% to 17.6% (z = 6.5, p < .001)
at 3 months. Twelve-month follow-up indicated a substantial reduction
in depressed mood with this reduction being more pronounced in those
reporting SI (mean (baseline) = 17.7 vs. mean (12 months) =
10.3) than in other patients (mean (baseline) = 11.1 vs. mean
(12 months) = 7.0).
"Conclusions
SI is common among individuals seeking CBMPs to treat a range of
chronic conditions and is associated with higher levels of depressed
mood and poorer quality of life. Treatment with CBMPs reduced the
prevalence and intensity of suicidal ideation."
https://www.tandfonline.com/doi/10.1080/13811118.2024.2356615?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed
[3866]
In "Controlled Inhalation of Tetrahydrocannabinol-Predominant
Cannabis Flos Mitigates Severity of Post-Traumatic Stress Disorder
Symptoms and Improves Quality of Sleep and General Mood in
Cannabis-Experienced UK Civilians: A Real-World, Observational Study"
from Sultan et al (2024):
"Results from this observational study suggest an association between
treatment with THC-predominant cannabis flowers and symptomatic
improvement for up to 6 months in a cohort of UK civilians diagnosed
with PTSD. The treatment was safe and well tolerated and characterized
by marked effects on quality of sleep, general mood, and severity of
PTSD-associated symptoms. Despite previous exposure to cannabis,
participants continued to report benefits after initiating treatment
with THC-predominant cannabis flowers."

https://karger.com/mca/article/7/1/149/912500/Controlled-Inhalation-of-Tetrahydrocannabinol
[3858]
In "The Effectiveness and Adverse Events of Cannabidiol and
Tetrahydrocannabinol Used in the Treatment of Anxiety Disorders in a
PTSD Subpopulation" (2023) from Stack et al of the University of
Sydney's School of Pharmacology and Applied Cannabis Research in
Sydney, NSW, Australia:
"The median doses taken were 50.0 mg/day for CBD and 4.4 mg/day for
THC. The total participant sample reported significantly improved
anxiety, depression, fatigue, and ability to take part in social roles
and activities. Those who were diagnosed with PTSD (n = 57) reported
significantly improved anxiety, depression, fatigue, and social
abilities. The most common AEs reported across the whole participant
cohort were dry mouth (32.6%), somnolence (31.3%), and fatigue
(18.5%), but incidence varied with different cannabis formulations.
The inclusion of THC in a formulation was significantly associated
with experiencing gastrointestinal AEs; specifically dry mouth and
nausea. Conclusions: Formulations of cannabis significantly improved
anxiety, depression, fatigue, and the ability to participate in social
activities in participants with anxiety disorders. The AEs experienced
by participants are consistent with those in other studies."
And of special note:
"Participants who took a THC-dominant formulation reported a
significant decrease in their anxiety levels. Those same participants
also represented the highest proportion of participants that were
classified as having clinical improvement (61.1%, n = 11), compared
with participants who were prescribed other formulation types. This
was unexpected as the median THC dose for this participant group was
33.8 mg/day, and it has been suggested that doses higher than 30
mg/day could be anxiogenic. A study reported that doses of CBD ranging
from 15 to 60 mg/day could offset the anxiogenic properties of THC,
which is reflected in our data; however, with lower doses of CBD
(median = 6.0 mg/day CBD)."
https://journals.sagepub.com/doi/pdf/10.1177/87551225231180796
[4557]
As assessed by Sultan et al (2024), "Controlled Inhalation of
Tetrahydrocannabinol-Predominant Cannabis Flos Mitigates Severity of
Post-Traumatic Stress Disorder Symptoms and Improves Quality of Sleep
and General Mood in Cannabis-Experienced UK Civilians: A Real-World,
Observational Study"
"Participants were asked to complete the PTSD checklist for civilians
(PCL-C). The PCL-C is a 17-item self-report measure of the 17 DSM-IV
symptoms of PTSD. The PCL-C has a variety of purposes, including
screening individuals for PTSD, diagnosing PTSD, and/or monitoring
symptom change during and after treatment. The PCL-C asks about
symptoms in relation to 'stressful experiences' and can be used with
any population. The symptoms endorsed may not be specific to just one
event, which can be helpful when assessing survivors who have symptoms
due to multiple events. Furthermore, the 17 items within the PCL-C can
be grouped into the four clusters of symptoms that characterize PTSD:
items 1-5 refer to symptoms of intrusion, questions 6 and 7 capture
symptoms of avoidance, questions 8-12 correspond to symptoms of
altered mood, and finally questions 13-7 report symptoms of altered
reactivity. Evidence suggests that a 5-10-point change represents
reliable change (i.e., change not due to chance) and a 10-20-point
change represents clinically significant change. Therefore, it is
recommended to use 5 points as a threshold for determining whether an
individual has responded to treatment and 10 points as a threshold for
determining whether the improvement is clinically meaningful."
And...
"Fifty-eight patients were included, 34 of which also had PROMs
[patient-reported outcome measures] recorded at 6 months. Most were
males (65.5%) with an average age of 39.2 years who had previously
used cannabis illicitly (95.6%). At 3 months, participants reported
significant improvements in overall health, mood, and sleep quality (p
< 0.001) but not in the proxy for HRQoL (p = 0.052). Similarly,
participants reported substantial benefits in managing intrusion
symptoms (p < 0.001), mood alterations (p < 0.001), and
reactivity alterations (p = 0.002), which were sustained or further
improved at 6 months. Participants did not report any side effects
associated with CBMPs."
https://pmc.ncbi.nlm.nih.gov/articles/PMC11521486/
[4906]
Ma et al (2025) looked at "Involvement of endocannabinoid receptor 1
in oxidative and inflammatory responses underlying anxiety-like
behavior in SPS&S-exposed mice":
"This study employed the single prolonged stress and shock (SPS&S)
model, a validated rodent paradigm of PTSD, to evaluate the expression
of key eCB system proteins within the medial preftontal cortex (mPFC),
hippocampus, and amygdala brain regions critically implicated in PTSD
pathophysiology (Ressler et al., 2022). Furthermore, using the
systemic administration of the CB1 receptor agonist WIN 55,212-2, we
investigated the modulation of CB1 receptor activation on oxidative
stress products, inflammatory responses and PTSD-related
behaviors."
...
"SPS&S exposure induced significant anxiety-like behaviors and
increased freezing in fear tests. It decreased CB1 receptor and
NAPE-PLD expression while increasing FAAH and MAGL levels in all
examined brain regions, alongside elevated IL-1β and caspase-1.
Plasma oxidative stress and inflammatory cytokines were also elevated.
WIN 55,212-2 administration mitigated anxiety-like behaviors but not
conditioned fear responses. Crucially, it normalized the elevated
plasma oxidative stress and inflammatory cytokine levels."
https://www.sciencedirect.com/science/article/abs/pii/S0014299925008143?via%3Dihub
[5306]
Of THC's endogenous homologue anandamide, Ney and Matthews (2025)
say:
"Posttraumatic stress disorder (PTSD) is characterized by profound
changes to stress systems and impairments in fear extinction, the
process believed to underlie recovery from a traumatic experience.
This chapter reviews the contribution of anandamide - an important
endocannabinoid - to human and animal stress responding as well as
fear conditioning with relevance to PTSD. Anandamide is critical to
the activation of the stress response and is differentially expressed
following exposure to trauma. It is also critical to the extinction of
conditional fear and may present a feasible target for treatment by
increasing the capacity for patients to consolidate extinction
memories during exposure therapy."
https://www.sciencedirect.com/science/article/abs/pii/B9780443190810000160
[3859]
Mederos et al (2024) elaborate on the role of the ECS in "Overwriting
an instinct: visual cortex instructs learning to suppress fear
responses":
"Fast instinctive responses to environmental stimuli can be crucial
for survival but are not always optimal. Animals can adapt their
behavior and suppress instinctive reactions, but the neural pathways
mediating such ethologically relevant forms of learning remain
unclear. We found that posterolateral higher visual areas (plHVAs) are
crucial for learning to suppress escapes from innate visual threats
through a top-down pathway to the ventrolateral geniculate nucleus
(vLGN). plHVAs are no longer necessary after learning; instead, the
learned behavior relies on plasticity within vLGN populations that
exert inhibitory control over escape responses. vLGN neurons receiving
input from plHVAs enhance their responses to visual threat stimuli
during learning through endocannabinoid-mediated long-term suppression
of their inhibitory inputs. We thus reveal the detailed circuit,
cellular, and synaptic mechanisms underlying experience-dependent
suppression of fear responses."
They explain:
"The model exhibited depression of inhibitory synapses onto vLGN
neurons that receive input from plHVAs during learning. Such a form of
synaptic plasticity, called long-term depression of inhibition (iLTD),
has been previously described in multiple brain areas in vitro and is
dependent on endocannabinoid (eCB) signaling. Heterosynaptic iLTD can
be triggered by activation of group I metabotropic glutamate receptors
(mGluR1 or mGluR5) in postsynaptic neurons. This causes release of
eCBs which act as retrograde messengers, activating eCB receptors
(CB1R) on nearby presynaptic inhibitory terminals which can induce a
long-lasting reduction of presynaptic GABA release probability. Allen
Institute gene expression data showed that eCB receptor CB1R and
mGluR5 are highly expressed in vLGN (Fig. S7A-B). We therefore
investigated if eCB-dependent iLTD in vLGN could mediate learned
suppression of escape."
https://www.biorxiv.org/content/10.1101/2024.07.31.605567v1.full.pdf
[4738]
In "Association of the HTR2A 102T/C polymorphism with attempted
suicide: a meta-analysis" Wang et al (2015) concluded:
"Our meta-analysis does not support the previously suggested
association between HTR2A 102T/C and attempted suicide in the general
population. However, in patients with schizophrenia, the C/C genotype
of 5-HT2A receptor 102T/C may increase the risk of attempted
suicide."
https://pubmed.ncbi.nlm.nih.gov/26075944/
[4742]
Turiaco et al (2024) ask about the "Genetics of suicide ideation. A
role for inflammation and neuroplasticity?" in the European Archives
of Psychiatry and Clinical Neuroscience:
"Exposure to suicide or suicide attempts increases the risk of
suicidal behavior. One of the strongest clinical predictors of SB is
the non-suicidal self-injury. SI and SB are favored by predisposing
risk factors such as loneliness, hopelessness, demoralization,
economics factors, cultural factors, or social isolation due to
belonging to a social minority."
Single-gene theories are now generally debunked.
"SCZ patients with SI have over a six-fold increase of suicide, while
people who had at least one lifetime psychotic event has double odds
of experiencing SI, triple odds of a future suicide attempt, and four
times the odds of dying by suicide. Given those data, the importance
of identifying biological and genetic biomarkers emerges. Our study
tried to reach this aim using a classic GWAS approach, a molecular
pathway analysis and modelling the polygenic risk score with the
clinical predictors in a model through machine learning. No SNP alone
reached a GWAS significance in identifying the genetic risk for
SI."
However...
"B3GALTL gene product codes for a protein that is implicated in
synaptogenesis. Quite interestingly, B3GALTL is expressed in the brain
by neurons and astrocytes (proteinatlas.org). It is also expressed in
T cells. This finding is then of particular relevance: Microglia are
the primary immune system cells in the central nervous system, and
they function like peripheral macrophages, releasing a multitude of
pro-inflammatory cytokines and chemokines. Post-mortem examination of
suicide patients shows an increased density of microglia in anterior
cingulate cortex, dorsolateral prefrontal cortex, and mediodorsal
thalamus regions. An increased microglial activation is identified by
PET in patients with SI. Microglial cells affect the
tryptophan-kynurenine pathway, increasing the production of neurotoxic
metabolites such as quinolinic acid, a NMDA agonist and creating an
imbalance in the glutamatergic system. Glutamate neurotransmission is
the background of a normal cognitive and emotional processing, so its
disruption can lead to increased impulsivity, depressed mood, and
suicidality. Consistently with this finding, another important
molecule involved in neuroinflammation is the 18-kDa Translocator
Protein (TSPO), whose expression was significantly increased in
patients with SI, most robustly in the regions of the anterior
cingulate cortex. Refer to Table 5, for previous relevant studies
about schizophrenia, neuroinflammation and SI."
https://pmc.ncbi.nlm.nih.gov/articles/PMC11422468/
[4743]
Several lines of evidence indicate that abnormalities in the
functioning of the central serotonergic system are involved in the
pathogenesis of depressive illness and suicidal behavior.
https://pubmed.ncbi.nlm.nih.gov/19224112/
[298]
https://pubmed.ncbi.nlm.nih.gov/29526601/
[299]
https://pubmed.ncbi.nlm.nih.gov/11727894/
[300]
Research does not seek to eliminate environmental influences in
favour of genetic disposition in depression, suicidal ideation and
completed suicide. But there are only two hypotheses for the high
incidence in Slovenia, both of which may be right, namely that some
are genetically predisposed to snap when stress is prolonged or
reaches a certain level, or that living conditions for some, according
to the rules constructed by their own society, are relentlessly
sub-optimal.
Slovenia is an ideal candidate for psychedelic therapy. Slovenia
ruminates endlessly about the past. Millions of tonnes of CO2 are
expended generating hot air about long-dead regimes and ideologies,
because they have affected people in this area. But arguing about what
Tito wrote about Stalin in 1940 won't bring your dead relatives back
or make you happy and strong today. To ignore the past and bury the
hatchet seems like a cop-out. But the past is getting in the way of
the present, and the future.
"In 1976, a man known as Ka-Tzetnik 135633 (original name: Yehiel
De-Nur) underwent LSD-assisted psychotherapy. At the time, he was
under the supervision of Prof. Jan Bastiaans at the State University
of Leiden in the Netherlands. Bastiaans was the psychiatrist who had
first identified concentration camp syndrome, also known as survivor
syndrome. This syndrome is a form of what we would now call
post-traumatic stress disorder (PTSD). Its characteristics include
social withdrawal, sleep disturbance, anxiety, and depression.
Bastiaans had also successfully treated this syndrome with
LSD-assisted psychotherapy. Ka-Tzetnik 135633 was a survivor of
Auschwitz, where he was a prisoner for two years. This trauma was
followed by three decades of torment, in which he relived the horror
of those two years at night. Through LSD-assisted psychotherapy,
however, Ka-Tzetnik 135633 found healing.
"Almost a half century after Bastiaans' successful trials, the
regulation of psychedelics for treating PTSD is finally underway.
Think of the Holocaust survivors who might have found healing through
these medicines, but couldn't because of a politically-motivated ban
on psychedelics. This realization brings home the horrific impact of
the failed war on drugs.'"
https://realitysandwich.com/psychedelics-and-judaism/
[1092]
Many diagnosed with PTSD are unimpressed by the medical response.
Modlin et al (2025) report on self-treatment strategies: cannabis and
psychedelics were the two most preferred, with MDMA a close third:
"An online survey recruited individuals with self-reported trauma
symptoms or a formal diagnosis of post-traumatic stress disorder
(PTSD)/complex post-traumatic stress disorder (CPTSD). Participants
were asked about their treatment history, satisfaction with current
treatments, and use of illicit substances for symptom management.
Further, after receiving psychoeducation on
3,4-methylenedioxymethamphetamine (MDMA) and psilocybin therapies,
participants' perceptions and willingness to participate in these
treatments were assessed.
"Results: Of the 873 respondents, 94.8% reported experiencing
psychological trauma, with 73.4% diagnosed with PTSD or CPTSD. Many
had attempted multiple treatments, predominantly medications and
various psychotherapies, but reported high dissatisfaction.
Significant rates of marijuana, psychedelics, and MDMA use for
self-management of trauma symptoms were reported, with minimal
physical and psychological complications. Willingness to try MDMA and
psilocybin therapies was high (0.81 and 0.83, respectively). Notably,
women and heterosexual individuals showed lower willingness, while
younger respondents and those with higher education levels showed
greater willingness to try these treatments.
"Conclusion: High willingness to try MDMA and psilocybin therapies
among trauma-exposed individuals highlights the need for further
research and clinical trials. Understanding demographic variations in
willingness can guide the development of accessible and effective
treatment options for PTSD and CPTSD. Public education about potential
risks and harm reduction strategies is crucial to promote safe and
informed use of these emerging therapies."
https://psychiatry-psychopharmacology.com/Content/files/sayilar/142/S3-S19.pdf
[5365]
Psychedelics break the rumination cycle by disrupting - in a good way
- brain activities which perpetuate habitual thinking and behaviour.
The most abundant 5-HT receptors expressed in the medial prefrontal
cortex (mPFC) are 5-HT1AR and 5-HT2AR. The 5-HT2AR pathway is enhanced
by 5-HT2AR-agonist psychedelics.
https://pubmed.ncbi.nlm.nih.gov/28858536/
[301]
In mice, given a single dose of psilocybin or LSD, and using the
social reward conditioned place preference (sCPP) assay Nardou et al
have made an important step forward in understanding the mechanism by
which psychedelics can override cultural log-jams retarding progress
and societal happiness, such as we see in Slovenia. "Psychedelics
reopen the social reward learning critical period" (2023) finds:
"Psychedelics are a broad class of drugs defined by their ability to
induce an altered state of consciousness. These drugs have been used
for millennia in both spiritual and medicinal contexts, and a number
of recent clinical successes have spurred a renewed interest in
developing psychedelic therapies. Nevertheless, a unifying mechanism
that can account for these shared phenomenological and therapeutic
properties remains unknown. Here we demonstrate in mice that the
ability to reopen the social reward learning critical period is a
shared property across psychedelic drugs. Notably, the time course of
critical period reopening is proportional to the duration of acute
subjective effects reported in humans. Furthermore, the ability to
reinstate social reward learning in adulthood is paralleled by
metaplastic restoration of oxytocin-mediated long-term depression in
the nucleus accumbens. Finally, identification of differentially
expressed genes in the 'open state' versus the 'closed state' provides
evidence that reorganization of the extracellular matrix is a common
downstream mechanism underlying psychedelic drug-mediated critical
period reopening. Together these results have important implications
for the implementation of psychedelics in clinical practice, as well
as the design of novel compounds for the treatment of neuropsychiatric
disease."
https://www.nature.com/articles/s41586-023-06204-3
[2825]
But we do not need to rely on mice when there are real-life mass
traumas going on. A special opportunity came along on 7 October 2023
in the Hamas attack on Supernova. "Trauma Under Psychedelics: MDMA
Shows Protective Effects During the Peritraumatic Period" say Netzer
et al (2024):
"Traumatic events (TEs) play a causal role in the etiology of
psychopathologies such as depression and posttraumatic stress disorder
(PTSD). Recent research has highlighted the therapeutic potential of
psychoactive substances and especially
3,4-methylenedioxymethamphetamine (MDMA), in alleviating trauma
symptoms in chronic patients. However, little is known regarding the
consequences of trauma that is acutely experienced under the influence
of psychoactive substances. Here we investigated the acute experiences
and peritraumatic processing of 657 survivors from the high-casualty
terror attack at the Supernova music festival in Israel on October
7th, 2023. Data were collected four to twelve weeks following the TE.
Approximately two-thirds of survivors were under the influence of
psychoactive substances at the time of the TE, offering a tragic and
unique natural experiment on the impact of psychoactive compounds on
TE processing. Our findings reveal that individuals who experienced
the trauma while under the influence of MDMA demonstrated
significantly improved intermediate outcomes compared to those who
were under the influence of other substances or no substances at all.
Specifically, the MDMA group reported increased feelings of social
support, more social interactions and enhanced quality of sleep during
the peritraumatic period, yielding reduced levels of mental distress
and reduced PTSD symptom severity. These novel findings suggest that
the influence of MDMA during the TE may carry protective effects into
the peritraumatic period, possibly mediated through the known effects
of MDMA in reducing negative emotions and elevating prosociality.
These protective effects in turn may mitigate the development of early
psychopathology-related symptoms. Current preliminary results
underscore the need for further understanding of the cognitive and
physiological processes by which psychedelic substances intersect with
trauma recovery processes."
https://www.biorxiv.org/content/10.1101/2024.03.28.587237v1
[3174]
With "Facing trauma under the influence of psychedelics: A
phenomenological study with Nova rave survivors" psychedelic research
found a new angle. Two thirds of the Israeli festivalgoers attacked On
7 October 2023 were under the influence of LSD, mushrooms, ketamine or
MDMA, say Simon et al (2025):

"Participants' awareness and knowledge about their substance use
created an 'epistemic container,' which may have facilitated real-time
containment of traumatic input while complicating later
meaning-making. A neurophenomenological pattern emerged wherein
psychedelic subjective effects appeared suppressed during the acute
trauma exposure, resurging after the threat was over. Quantitative
analysis showed a predominantly positive subjective impact of
substance use on immediate survival (75%-79%) and emotional coping
(83%-84%) but mixed outcomes in aftermath processing (42%-53% positive
and 25%-26% negative).
"Conclusions:
Psychedelic-induced dissociation during trauma exposure may confer
acute adaptive benefits while causing integration challenges a paradox
with significant implications on trauma research. This singular
perspective on the psychedelic-trauma interface invites further
research into these complex neuropsychological interactions.
Psychedelics may serve as epistemological instruments, revealing
cognitive processes and unique prisms through which trauma response,
psychopathology, resilience, and the adaptive capacities of the human
mind under extreme conditions can be re-examined."
Unfortunately evaded in this "unique interaction between psychedelic
states and extreme stress" (a description suggesting the authors have
not studied tripping in Lincoln on a Saturday night) was the
opportunity to include a control group.
So the chances of comparing survival rates and other parameters
between users and non-users have been lost.
With this methodology there was no danger of demonstrating
evolutionary advantages of elevated consciousness, although the
authors findings suggest exactly this: psychedelics use "seems to have
created a distinct form of psychological protection that both aided
immediate survival and posed challenges for later integration".
"Optimistic detachment" helped users to cope with the threat and being
sharp enabled them to act smart.
How to help never-users understand this? In support of the Defence
claim that the ZPPPD as it applies to psychedelics (excluding ketamine
and MDMA) is an unconstitutional restriction of evolutionary
advantage, the Defendant compares the value of psychedelics amid
stressful conditions to the "unconscious competence" of driving a
vehicle normally (second-nature behaviour, background-task capable),
as opposed to sweating neurotically over every control movement.
As for any increased difficulties in the aftermath, you remember the
last book you read, but are unlikely to recall any specific text. You
won't have to worry about PTSD recovery later if you are dead. If you
have more information to integrate, chances are the result will be
more integrated.
Once again, the opportunity to compare recovery patterns between
psychedelic and non-users with controls was eluded rather than
elucidated in this, perhaps the ultimate survivor-biased study.
Perhaps the other third of the festivalgoers couldn't be found.
https://journals.sagepub.com/doi/10.1177/02698811251372508?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed
[5651]
As of 2025 no comparison of survival by drug type at Supernova
pertains. However, the Court must hear that it is more bad news for
any followers of Slovenian culture who find themselves under attack
as, following a study by Nacasch et al (2025):
"The team hypothesised that survivors who were on drugs at the time
would have been more hyper-aware of the event and their surroundings,
thereby experiencing more post/trauma from the attack overall.
However, they were wrong."
Unfortunately for Slovenia's majority drug-choosers:
"'It was the alcohol,' [Professor Mark Weiser] told Health Policy
Watch. 'These people were the ones who had more severe post-traumatic
symptoms.'"
https://healthpolicy-watch.news/study-alcohol-not-psychedelics-linked-to-heightened-trauma-in-survivors-of-attack-on-israeli-festival-goers/
[5652]
Note: this was one of those occasions when alcohol wasn't a "drug".
"Seventy-one of them (57.7%) reported using psychoactive drugs at the
festival - 12 only alcohol, nine only lysergic acid (LSD), seven only
3,4-methylenedioxymethamphetamine (MDMA), six only cannabis, three
only methylmethcathinone (MMC), 15 various drug combinations including
alcohol, and 19 various drug combinations excluding alcohol."
Despite all the harmful illegality adding to the background stress,
the pro-alcohol researchers had to admit defeat:
"The multiple regression analysis of the PDEQ [Peritraumatic
Dissociative Experiences Questionnaire] dissociation score with VAS-A
[Visual Analog Scale for Anxiety], GAD-7 [Generalized Anxiety
Disorder-7], PHQ-9 [(Patient Health Questionnaire-9], and PDS-5
[Posttraumatic Diagnostic Scale] subscale scores, including gender and
age, was statistically significant (p=0.0001)."
Each of the component scores were also statistically significant
except VAS-A. In more great news for the nation's psychiatrists and
pharma concerns...
"...in marked contrast to our expectations, we found that only
pre-trauma alcohol consumption, with or without other drugs,
significantly increased the risk of peri-traumatic dissociation,
anxiety,depression, and ASD [acute stress disorder] symptoms."
https://onlinelibrary.wiley.com/doi/pdf/10.1002/wps.21254
[5653]
Another alternative to mice is "Human pluripotent stem cells as a
translational toolkit in psychedelic research in vitro". As Salerno
and Rehan explain:
"PSCs can differentiate into various brain cell types, mirroring
endogenous expression patterns and cell identities to recreate disease
phenotypes. Brain organoids derived from PSCs resemble cell diversity
and patterning, while region-specific organoids simulate circuit-level
phenotypes. PSC-based models hold significant promise to illuminate
the cellular and molecular substrates of psychedelic-induced
phenotypic recovery in neuropsychiatric disorders."
and
"The identities of the signaling pathways responsible for
psychedelics' hallucinogenic and therapeutic properties and whether
these are distinct or overlapping pathways are some of the questions
still under debate.
"The Gq or β-arrestin-2 recruitment paradigm represents a
glimpse into the extensive network of pathways regulated by 5-HT2AR
activation via psychedelics. For example, distinct psychedelics
activate phospholipase A2 and phospholipase D via ADP-ribosylation
factor 1. The activation of 5-HT2AR, triggered by various agonists
including DOI and LSD, can lead to the heterodimerization of the
receptor with metabotropic glutamate receptor 2 and dopamine D2
receptor, both of which are Gi-coupled receptors. This transactivation
blocks the cAMP's synthesis by adenylate cyclase and prompts
heterotrimeric Gi/o proteins to trigger Src-mediated downstream
events.
"A burst of glutamate also follows psychedelic administration, mainly
in the cortical layer V of the neocortex, which is a 5-HT2AR-enriched
area.8,23 High glutamate levels activate the
α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor
(AMPAR), boosting the secretion of brain-derived neurotrophic factor
(BDNF), which in turn signals through the tyrosine receptor kinase B
(TrkB) and mammalian target of rapamycin (mTOR) pathways, sustaining
both the AMPAR activation and BDNF secretion in a positive feedback
loop of neural plasticity.
"Furthermore, the hallucinogenic properties of different psychedelics
are also proposed to be influenced by other 5-HTRs, and not all
5-HT2AR agonists have hallucinogenic properties. For instance, human
studies also provide evidence supporting the involvement of the
5-HT1AR in the effects of psilocybin. Competition binding studies on
rodent brains and primary cells revealed a lack of pronounced
selectivity of psilocin for 5-HT2AR over 5-HT1AR. Nevertheless, the
role of the 5-HT2AR remains the subject of more extensive study and
investigation. Ketanserin, a selective antagonist of the 5-HT2Rs,
effectively eliminates head-twitch behavioral responses in mice;
however, it does not attenuate psilocybin-induced structural
modifications in the prefrontal cortex. As a result, uncertainties
remain about whether and how the neuroplastic effects on rescuing
disease phenotypes relate to behavioral responses, especially in
humans. Moreover, human studies investigating the molecular mechanisms
of psychedelic-induced plasticity rely on global parameters such as
peripheral BDNF levels, which may not correlate with the molecular
mechanisms occurring in brain cells. For in-depth cellular and
molecular insights into the role of 5-HT2AR, in vitro investigations
are required."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11019282/
[3173]
Using PSCs Schmidt et al (2024) of the University of Heidelberg
showed that psilocin causes 5-HT2A receptor internalization and
redistribution in human cortical neurons, induces BDNF expression and
downstream signaling in human neurons in a dose-dependent manner,
induces gene expression changes associated with axonal and synaptic
plasticity, increases dendritic complexity, and increases synaptic
strength and synaptogenesis.
https://www.researchgate.net/publication/381253376_Psilocin_fosters_neuroplasticity_in_iPSC-derived_human_cortical_neurons
[3175]
Blest-Hopley et al (2025) found "improved mental health outcomes and
normalised spontaneous EEG activity in veterans reporting a history of
traumatic brain injuries following participation in a psilocybin
retreat":
"This study investigates the effects of psilocybin administered in
retreat settings on veterans with a history of TBI, focusing on mental
health outcomes and changes in brain connectivity as measured by
EEG.
"Methods: A total of 21 participants were recruited through the
Heroic Hearts Project, which facilitated access to two six-day
psilocybin retreats in Jamaica. Before the retreat, participants
underwent three individual and three group coaching sessions to
prepare for the experience. During the retreat, two psilocybin
ceremonies were held, spaced 48 hours apart. Participants received an
initial dose of 1.5g to 3.5g of dried psilocybin mushrooms, with the
option to increase the second dose up to 5g. Psilocybin was
administered in a tea format, under the supervision of experienced
facilitators. Psychological outcomes were assessed using validated
questionnaires (PCL-5, PHQ-9, STAI) at baseline (four weeks
pre-retreat) and four weeks post-retreat. Electroencephalography (EEG)
was used to measure brainwave activity pre- and post-treatment. Paired
t-tests were used to analyze changes in psychological scores, while
EEG frequency band analysis assessed changes in brain function and
connectivity.
"Results: Improvements were observed across several mental health
measures: PTSD (PCL-5 scores decreased by 50%, p=0.010), depression
(PHQ-9 scores decreased by 65%, p<0.001), and anxiety (STAI) scores
decreased by 28%, p<0.001). EEG data showed decreased delta and
theta power in frontal and temporal regions, indicating potential
improvements in cognitive control and emotional processing. Enhanced
coherence in alpha and beta bands suggested improved neural
communication."
https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2025.1594307/full
[5324]
Administration of psychedelics to a relatively small number of people
could bring the paralysing stand-off over Slovenia's past to a quick
and elegant halt. The participants would see the damage they are doing
- to themselves and the country - by perpetuating hostilities against
groups and against individuals long dead. They would be able to more
accurately gauge the importance of the historical against the
challenges of the present and future. They would be less susceptible
to fearmongering and superstition. Their nightmares would be put into
perspective.
The Slovenians who stand to gain the most from ranting on about the
past for political purposes are the least likely to have taken them.
Among them you will find the most enthusiastic opponents of the
benefits of psychedelics, about which their ignorance resembles what
the average villager in Radenci knows of Tito in the 1940s, i.e. what
they have been told, not what they have experienced.
Unnecessary and counterproductive rigidity are legal and mental
constructs that can be ameliorated by mind-expanding drugs. They offer
a sideways view, and offer users the opportunity to invite unnecessary
and counterproductive rigidity to get lost. And this is why the
anti-drug religion, prohibition, doesn't like them.
Oblivion from alcohol, often associated with violence, is a legally
approved coping mechanism, but is solipsistic and does nothing to
solve the underlying malaises. Drugs other than alcohol, which can
give rise to creative solutions to life stresses or at least relief by
way of euphoria are legally disapproved, and the users accused of
escapism. A drug user is not concerned with his or her genetic
predisposition. They can't change that.
What Slovenia's warring factions need is not more press stories about
what the communists did, but consolation now. Now does not contain the
grudges of the past, nor the anxieties of the future. Now is not a
popular concept for political manipulators. "Now" suggests something
should be done, rather than talked about. "Now" cannot play on
people's resentments and fears. Dealing with "Now" may be financially
expensive compared to promises and grumbling.
Although it is not necessary to understand the mechanisms of the
human brain by which consolation takes place, and psychedelic people
have managed without this so far, it's good to know the scientists are
finally being allowed to fill in the details, and the activation of
5-HT1A receptors in the anterior cingulate cortex was sufficient to
reverse consolation and sociability deficits in Chinese mandarin
voles.
https://elifesciences.org/articles/67638
[302]
I think it might work in Slovenians too.
From "The Role of Nutrients in Protecting Mitochondrial Function
and
Neurotransmitter Signaling: Implications for the Treatment of
Depression, PTSD, and Suicidal Behaviors" by Du et al (2016):
"Drugs, which lower the cholesterol levels, are able to cause the
depressive symptoms (Tatley and Savage, 2007). In addition,
fish-intake (rich in EPA or DHA) directly protects against the onset
of major depressive disorder (Weidner et.al., 1992). Moreover, chronic
stress induced an inhibition to the respiratory chain in the
mitochondria in the brain (Madrigal et al., 2001). Mitochondrial
dysfunction caused by changes in biochemical cascade or the damage to
the mitochondrial electron transport chain has been suggested to be an
important pathogenic factor for the psychiatric disorders,
particularly in bipolar disorders and depression (Rezin et al., 2009).
Moreover, food supplements, such as B12 or folate, which protects
mitochondrial functions are effective as an adjunctive therapy for the
treatment of depression (Papakostas et al., 2005). All these studies
imply that mitochondrial dysfunction and reduced formation of lipid
raft may be involved in the etiology of depression and suicidal
behavior (Figure 1). These stress-induced neuronal dysfunctions
interact with the other genetic and environmental factors, including
adverse childhood events, exposure to trauma, drug abuse, smoking,
alcohol usage, sleep, diet, and exercise levels, to precipitate mood
disorders in genetically and/or physiologically vulnerable or
predisposed individuals (Figure 1)."
As for suicide:
"Epidemiological studies have identified low fish (high in ω-3
fatty acid) consumption as a risk factor for mortality from suicide
(Hibbeln and Salem, 1995, Hirayama, 1990, Sublette et al., 2006,
Tanskanen et al., 2001). One study noted that frequent fish
consumption (twice per week or more) significantly reduced the risk of
depressive symptoms and of self-reported suicidal ideation (Tanskanen
et al., 2001). A 17-year follow-up study of over 250,000 Japanese
subjects showed that people who ate fish daily had a lower risk of
death from suicide (Hirayama 1990). In addition, several reports
indicate that lower ω3-fatty-acid levels, including lower plasma
EPA, and DHA, or EPA in red blood cells, predicted greater risk of
suicide attempt (Hibbeln and Salem, 1995, Huan et al., 2004, Sublette
et al., 2006). Because both cholesterol and DHA are major components
of the lipid raft, it is possible that reduced cholesterol and
ω3-fatty-acid levels may affect the formation of lipid rafts in
the CNS, and subsequently reduce neurotransmitter signaling (Czysz and
Rasenick, 2013). Notably, increased formation of lipid rafts in the
membrane would facilitate serotonergic (Donati et al., 2008, Renner et
al., 2007), dopaminergic (Villar et al., 2009), and glutamatergic
(Francesconi et al., 2009, Ponce et al., 2008) neurotransmitter
signaling; all of these play important roles in the pathophysiology
and treatment of psychiatric disorders. Studies have noted that low
cholesterol levels are associated with increased risk of suicide
(Neaton et al., 1992) and that this association shows an inverse
relationship with baseline total serum cholesterol (Lester, 2002,
Lindberg et al., 1992). Other studies found that individuals who
attempted suicide had significantly lower cholesterol levels than
controls (Atmaca et al., 2002, Boston et al., 1996, Kim et al., 2002,
Kunugi et al., 1997, Maes et al., 1997a, Modai et al., 1994,
Rabe-Jablonska and Poprawska, 2000, Sarchiapone et al., 2001, Takei et
al., 1994). A postmortem study found that the brains of violent
suicide completers had a lower grey-matter cholesterol content
(Lalovic et al., 2007), and that a family history of suicidal behavior
was more frequent among carriers of Smith-Lemli-Opitz syndrome, an
autosomal recessive disorder characterized by abnormally low
cholesterol levels (Lalovic et al., 2004)."

https://europepmc.org/backend/ptpmcrender.fcgi?accid=PMC4417658&blobtype=pdf
[1817]
By 2023, Song et al's highlights were
"Mitochondrial dysfunction plays a vital role in the etiology of
depression.
Dysregulation of the mitochondrial quality control system exacerbates
the pathophysiology of depression.
Mitochondrial energy metabolism disorders fail to provide
physiological support for neuroplasticity in depression.
The interaction between defective mitochondria and neuroinflammation
worsens depression.
Mitochondria represent a potential target for pharmacological
intervention of depression."

We are reminded:
"BDNF, a substance released by the nerve growth factor family, known
as neurotrophins, controls neuronal survival, preserves neuronal
function and synaptic plasticity, and promotes the synthesis and
release of neurotransmitters . BDNF plays a neuroprotective role by
increasing the respiratory control index via the mitogen-activated
protein kinase (MEK)/Bcl2. Therefore, suppression of the Bcl2-related
signaling pathway reduces BDNF levels, which consequently affects
mitochondrial function, resulting in the impairment of plasticity and
initiation of apoptosis. In addition to manufacturing oxygen and
nitrogen species for synaptic plasticity, mitochondria trigger
caspases in dendrites to eliminate postsynaptic spines associated with
chronic depression. In line with mitochondrial function, BDNF enhances
the utilization ratio of oxidation to enable neuronal
plasticity."
https://www.sciencedirect.com/science/article/pii/S0753332223014506
[3998]
According to a 2020 review in BMC Psychiatry by Raymundi et al at the
Federal University of Parana in Brazil:
"At low doses, THC can enhance the extinction rate and reduce anxiety
responses. Both effects involve the activation of cannabinoid type-1
receptors in discrete components of the corticolimbic circuitry, which
could counterbalance the low 'endocannabinoid tonus' reported in PTSD
patients. The advantage of associating CBD with THC to attenuate
anxiety while minimizing the potential psychotic or anxiogenic effect
produced by high doses of THC has been reported. The effects of THC
either alone or combined with CBD on aversive memory reconsolidation,
however, are still unknown.
"Conclusions: Current evidence from healthy humans and PTSD patients
supports the THC value to suppress anxiety and aversive memory
expression without producing significant adverse effects if used in
low doses or when associated with CBD. Future studies are guaranteed
to address open questions related to their dose ratios, administration
routes, pharmacokinetic interactions, sex-dependent differences, and
prolonged efficacy."

"As reviewed here, low doses of THC attenuate aversive memory
expression through anxiety reduction, extinction facilitation, and
reconsolidation impairment (currently shown in laboratory animals
only."
https://bmcpsychiatry.biomedcentral.com/counter/pdf/10.1186/s12888-020-02813-8.pdf
[1506]
By 1985 Howlett had shown "Cannabinoid inhibition of adenylate
cyclase. Biochemistry of the response in neuroblastoma cell
membranes". The fight-or-flight response, which is triggered by
adrenaline, relies on cAMP signaling to increase heart rate and
mobilize energy stores. This system is hyperactive and triggered too
easily in PTSD. Inhibiting adenylyl cyclase would impair this
response.
https://chemport-n.cas.org//chemport-n/?APP=ftslink&action=reflink&origin=npg&version=1.0&coi=1%3ACAS%3A528%3ADyaL2MXhvFOqsL0%3D&md5=49106efb644a6fc89de96c071d7d766b
[3731]
By 1997 Glass et al had shown using receptor audioradiography
that
"...cannabinoid receptor binding sites in the human brain are
localized mainly in: forebrain areas associated with higher cognitive
functions; forebrain, midbrain and hindbrain areas associated with the
control of movement; and in hindbrain areas associated with the
control of motor and sensory functions of the autonomic nervous
system."
https://pubmed.ncbi.nlm.nih.gov/9472392/
[3729]
Marsicano et al published "The endogenous cannabinoid system controls
extinction of aversive memories" in 2002:
"Acquisition and storage of aversive memories is one of the basic
principles of central nervous systems throughout the animal kingdom.
In the absence of reinforcement, the resulting behavioural response
will gradually diminish to be finally extinct. Despite the importance
of extinction, its cellular mechanisms are largely unknown. The
cannabinoid receptor 1 (CB1) and endocannabinoids are present in
memory-related brain areas and modulate memory. Here we show that the
endogenous cannabinoid system has a central function in extinction of
aversive memories. CB1-deficient mice showed strongly impaired
short-term and long-term extinction in auditory fear-conditioning
tests, with unaffected memory acquisition and consolidation. Treatment
of wild-type mice with the CB1 antagonist SR141716A mimicked the
phenotype of CB1-deficient mice, revealing that CB1 is required at the
moment of memory extinction. Consistently, tone presentation during
extinction trials resulted in elevated levels of endocannabinoids in
the basolateral amygdala complex, a region known to control extinction
of aversive memories. In the basolateral amygdala, endocannabinoids
and CB1 were crucially involved in long-term depression of GABA
(gamma-aminobutyric acid)-mediated inhibitory currents. We propose
that endocannabinoids facilitate extinction of aversive memories
through their selective inhibitory effects on local inhibitory
networks in the amygdala."
https://pubmed.ncbi.nlm.nih.gov/12152079/
[3730]
With understanding of the glucocorticoid mechanism in mammals
somewhat stuck, in 2003 Di et al revealed "Nongenomic Glucocorticoid
Inhibition via Endocannabinoid
Release in the Hypothalamus: A Fast Feedback Mechanism":
"The glucocorticoid effect was not blocked by the nitric oxide
synthesis antagonist NG -nitro-L-arginine methyl ester hydrochloride
or by hemoglobin but was blocked completely by the CB1 cannabinoid
receptor antagonists AM251
[N-(piperidin-1-yl)-5-(4-iodophenyl)-1-(2,4-dichlorophenyl)-
4-methyl-1H-pyrazole-3-carboxamide] and AM281
[1-(2,4-dichlorophenyl)-5-(4-iodophenyl)-4-methyl-N-4-morpholinyl-1H-pyrazole3-carboxamide]
and mimicked and occluded by the cannabinoid receptor agonist
WIN55,212-2 [( )-( )-[2,3-dihydro-5-methyl-3-(4-
morpholinylmethyl)pyrrolo[1,2,3-de]-1,4-benzoxazin-6-yl]-1-naphthalenylmethanone
mesylate], indicating that it was mediated by retrograde
endocannabinoid release."
https://www.jneurosci.org/content/jneuro/23/12/4850.full.pdf
[3733]
In 2009 new light was shed on the rapid effects of glucocorticoids in
mammals by revealing a putative nongenomic role of glucocorticoids in
regulating emotional learning in rodents through a coordinated
induction of endocannabinoid signaling. Campolongo et al showed
"Endocannabinoids in the rat basolateral amygdala enhance memory
consolidation and enable glucocorticoid modulation of memory":
"Extensive evidence indicates that the basolateral complex of the
amygdala (BLA) modulates the consolidation of memories for emotionally
arousing experiences, an effect that involves the activation of the
glucocorticoid system. Because the BLA expresses high densities of
cannabinoid CB1 receptors, the present experiments investigated
whether the endocannabinoid system in the BLA influences memory
consolidation and whether glucocorticoids interact with this system.
The CB1 receptor agonist WIN55,212-2 (5-50 ng per 0.2 μL per side),
infused bilaterally into the BLA of male Sprague-Dawley rats
immediately after inhibitory avoidance training, induced
dose-dependent enhancement of 48-h retention. Conversely, the CB1
receptor antagonist AM251 (0.07-0.28 ng per 0.2 μL per side)
administered after training into the BLA induced inhibitory avoidance
retention impairment. Furthermore, intra-BLA infusions of a low and
nonimpairing dose of AM251 (0.14 ng per 0.2 μL per side) blocked
the memory enhancement induced by concurrent administration of
WIN55,212-2. Delayed infusions of WIN55,212-2 or AM251 administered
into the BLA 3 h after training or immediate posttraining infusions of
these drugs into the adjacent central amygdala did not significantly
alter retention performance. Last, intra-BLA infusions of a low and
otherwise nonimpairing dose of AM251 (0.14 ng per 0.2 μL per side)
blocked the memory-enhancing effect induced by systemic administration
of corticosterone (3 mg/kg, s.c.). These findings indicate that
endocannabinoids in the BLA enhance memory consolidation and suggest
that CB1 activity within this brain region is required for enabling
glucocorticoid effects on memory consolidation enhancement."

https://www.pnas.org/doi/10.1073/pnas.0900835106
[3732]
In "Cannabinoid modulation of corticolimbic activation to threat in
trauma-exposed adults: a preliminary study" (2020) Rabinak et al at
Wayne State University in Detroit, explained:
"Excessive fear and anxiety, coupled with corticolimbic dysfunction,
are core features of stress- and trauma-related psychopathology, such
as posttraumatic stress disorder (PTSD). Interestingly, low doses of
Δ9-tetrahydrocannabinol (THC) can produce anxiolytic effects,
reduce threat-related amygdala activation, and enhance functional
coupling between the amygdala and medial prefrontal cortex and
adjacent rostral cingulate cortex (mPFC/rACC) during threat processing
in healthy adults. Together, these findings suggest the cannabinoid
system as a potential pharmacological target in the treatment of
excess fear and anxiety. However, the effects of THC on corticolimbic
functioning in response to threat have not be investigated in adults
with trauma-related psychopathology."
The results of their fMRI study of 71 human subjects undergoing a
well-established threat processing paradigm demonstrated that:
"In adults with PTSD, THC lowered threat-related amygdala reactivity,
increased mPFC activation during threat, and increased mPFC-amygdala
functional coupling."
...which suggests that
"THC modulates threat-related processing in trauma-exposed
individuals with PTSD..."
Taking fear and happiness as opposing states, the Defence applauds
the astonishing diligence with which scientists are working towards
the discovery of what cannabis users already know: it helps you relax,
reduces rumination, and makes you happy. These detailed, tiny steps
are important. But the backstory does not predate the legally relevant
ordinances of international cannabis control. This provides no useful
result today for the PTSD victim or recreational user, for whom these
effects are as intuitively obvious as those of coffee and do not
require scientific proof or peer review.
"Interestingly, CB1Rs are highly abundant in these regions of the
corticolimbic system (Tsou et al. 1998; Marsicano and Lutz 1999; Patel
et al. 2017) and recent neuroimaging studies in healthy adults show
that THC modulates activity in the amygdala and mPFC/rACC.
Specifically, an acute oral low dose of THC (7.5 mg) reduced amygdala
reactivity, but enhanced coupling between the amygdala and mPFC/rACC,
to social threat (i.e., fearful and angry facial expressions), as
compared to placebo (PBO; Phan et al. 2008; Gorka et al. 2015a). These
findings suggest that pharmacological enhancement of ECB signaling may
help to address corticolimbic dysfunction in PTSD and other
stress-related disorders. However, it should be noted that others have
reported that administration of THC, particularly at a higher dose (10
mg) increases amygdala activation (Bhattacharyya et al. 2010, 2017),
and modulates activation in frontal and parietal regions (Crippa et
al. 2009), while increasing levels of anxiety and autonomic arousal to
fearful faces (Crippa et al. 2009; Bhattacharyya et al. 2010, 2017).
These divergent findings highlight the complexity of THC's effect on
threat responding that may be bimodal, such that low doses of THC may
be anxiolytic (Wachtel et al. 2002), whereas higher doses of THC are
typically anxiogenic (D'Souza et al. 2004; Genn et al. 2004; Viveros
et al. 2005b; Bhattacharyya et al. 2015, 2017). To-date, the effects
of THC on corticolimbic responses to threat have only been conducted
in healthy individuals. Thus, it is unclear whether the observed
effects of THC on corticolimbic function and functional connectivity
would replicate in individuals with trauma exposure who are at risk
for developing PTSD and/or meet criteria for PTSD."
and with reckless abandon as to how their findings might be applied
in law to the anti-happy brigade who profit from the miseries of
prohibition, they conclude:
"Consistent with previous findings in healthy adults, we found that,
within the PTSD group, THC attenuated amygdala activation, increased
mPFC/rACC activation, and increased corticolimbic functional
connectivity to threat compared to PBO (Phan et al. 2008; Gorka et al.
2015)."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7244361/
[1507]
In "The Long-Term, Prospective, Therapeutic Impact of Cannabis on
Post-Traumatic Stress Disorder" by Bonn-Miller et al (2022)
decided:
"Given the increasing availability and use of cannabis among
individuals with post-traumatic stress disorder (PTSD) and the
addition of PTSD as an eligible diagnosis in several U.S. medical
cannabis programs [24 states as of 2020], the efficacy of
dispensary-obtained cannabis needs to be thoroughly examined."
and to the extent that they were able to do so...
"This prospective study [see 2362 page 332 for definition] assessed
PTSD symptoms and functioning every 3 months over the course of a year
in two samples of participants diagnosed with PTSD: (1) those with
PTSD using dispensary-obtained cannabis (cannabis users) and (2) those
with PTSD, who do not use cannabis (controls). Linear mixed-effects
models and generalized estimating equations tested whether
trajectories of symptoms differed between the two subsamples."
And so
"A total of 150 participants (mean [standard deviation] age, 50.67
[15.26] years; 73% male) were enrolled in the study. Over the course
of 1 year, the cannabis users reported a greater decrease in PTSD
symptom severity over time compared to controls [group×time
interaction=−0.32 (95% confidence interval [CI]=−0.59 to
−0.05, R2=0.13; t=−2.35, p=0.02). Participants who used
cannabis were 2.57 times more likely to no longer meet DSM-5 criteria
for PTSD at the end of the study observation period compared to
participants who did not use cannabis (95% CI=1.12-6.07;
p=0.03)."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9070744/
[1509]
In "Anxiety severity and prescription medication utilization in
first-time marijuana users" from Dugosh et al (2023):
"The study sample was comprised of 108 adults with anxiety or PTSD as
a referring condition; they were enrolled in a longitudinal study
evaluating biopsychosocial outcomes in new MM patients. Consenting
participants completed an assessment battery at baseline and Month 3
(n = 94, 87 % follow-up rate) that included a measure of anxiety
severity and questions about current anxiety medication prescription
and desired (baseline) and actual (Month 3) reductions in medication
use.
"Results
Findings indicated that 59 % of participants reported prescription
medications for anxiety, with 70 % reporting at least a moderate
desire to reduce medication use. Overall and within the medication
sub-sample, participants displayed significant reductions in anxiety
severity from baseline to Month 3 (p's <0.0001). Furthermore, 32 %
reported actual reductions in medication use at Month 3, and
reductions were more likely among patients prescribed benzodiazepines
than other drug classes.
"Conclusions
Results suggest that a significant number of MM patients with anxiety
and/or PTSD diagnoses are currently being prescribed antianxiety
medications and that MM may help to reduce their use of these
medications.
"Limitations
Limitations include the observational study design and the lack of a
PTSD-specific measure. More controlled longitudinal studies are
necessary to better understand the role of MM in the treatment of
anxiety and PTSD."
https://www.cannabisclinicians.org/2023/10/27/anxiety-severity-and-prescription-medication-utilization-in-first-time-medical-marijuana-users-2/
[4190]
Again, in 2023, Vaddiparti et al found
"PTSD symptom severity as measured by total PCL-5 score improved
significantly at 30- and 70-day follow-ups. Similarly, statistically
significant reductions in nightmares were reported at 30- and 70-day
follow-ups. Corresponding improvements in sleep were noticed with
participants reporting increased duration of sleep hours, sleep
quality, sleep efficiency, and total PSQI score. Likewise, negative
affect and global mental health improved significantly at follow-up.
According to the post hoc analyses, the most statistically significant
changes occurred between baseline and 30-day follow-up. The exception
to this pattern was nightmares, which did not show significant
improvement until day 70."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10642978/
[4254]
In 162 patients with PTSD identified from the UK Medical Cannabis
Registry, tested with Impact of Events Scale-Revised (IES-R),
EQ-5D-5L, Single-Item Sleep Quality Scale (SQS), Generalized Anxiety
Disorder-7 (GAD-7) and Patient Global Impression of Change (PGIC),
Pillai et al (2022) found that:
"Statistically significant improvements were seen in all domains of
the IES-R, and both GAD7 and SQS, at all lengths of follow-up (p <
0.050). The EQ-5D-5L index score and both 'usual activities' and
'anxiety and/or depression' subscales also improved significantly at
all follow-ups (p < 0.050). Significant improvements in the
'self-care' and 'pain and discomfort' subscales were seen at 1- and
3-month follow-up and the 'mobility' subscale at 3-months only (p <
0.050). There was a large effect size (r = 0.50) for the IES-R
hyperarousal score at 3-month follow-up. All other IES-R and GAD7
scores had medium effect sizes. PGIC1 median scores were consistently
6.00 (5.00–6.00) at 1-, 3-, and 6-month follow-up, whilst median
PGIC2 was 3.00 (2.00–3.00) at 1- and 3-month follow-up, and 2
(1.00–3.00) at 6-months (n = 128, 93, and 51
respectively)."
https://www.tandfonline.com/doi/full/10.1080/14737175.2022.2155139 [4694]
Mayo et al, reviewing the issue of "Targeting the Endocannabinoid
System in the Treatment of Posttraumatic Stress Disorder: A Promising
Case of Preclinical-Clinical Translation?" in 2022, say:
"Preclinical research shows that eCB activity influences functional
connectivity between the prefrontal cortex and amygdala and thereby
influences an organism's ability to cope with threats and stressful
experiences. Animal studies show that CB1 receptor activation within
the amygdala is essential for extinction of fear memories. Failure to
extinguish traumatic memories is a core symptom of posttraumatic
stress disorder, suggesting that potentiating eCB signaling may have a
therapeutic potential in this condition. However, it has been unknown
whether animal findings in this domain translate to humans. Data to
inform this critical question are now emerging and are the focus of
this review."
Under the headling "Fear" they say
"The loss-of-function FAAH 385A allele was inserted into mice,
resulting in decreased FAAH activity and concomitantly increased AEA
levels. In mice and humans, carriers of the A-allele had enhanced
fronto-amygdala connectivity and fear extinction. We subsequently
reported similar effects in humans, including a gene dose-dependent
effect of the A-allele on peripheral AEA levels. Individuals
homozygous for the A-allele showed enhanced fear extinction and recall
of extinction learning when tested 24 hours later. These findings have
since been replicated by others, showing that fear extinction is
related to peripheral AEA levels."
And the latest scientists to discover cannabis think...
"The available human data consistently support a translation of
animal findings on fear memories and stress reactivity and suggest a
potential therapeutic utility in humans."
Their Figure 1 shows the approaches to ECS pathways from a drug
manufacturing perspective

https://www.sciencedirect.com/science/article/pii/S0006322321014724
[4010]
Taking another overview of the evidence in 2023, Marchetta et al
"...describe the three fear memory phases (e.g., consolidation,
reconsolidation and extinction) that are most significantly altered in
PTSD patients, and...review data from both human and animal studies on
the effects of repeated exposure to trauma reminders on fear memory
retention and extinction combined with pharmacological
intervention."
Highlights include
"Based on preclinical studies, the monoamine hypothesis of PTSD has
been enlarged with a more complex neurochemical and neuroplasticity
hypothesis that highlights the role of the glutamatergic system in
trauma and stress psychopathology."
"The mechanisms sustaining systems consolidation have not been fully
elucidated."
As for reconsolidation
"...differently to what the name might suggest, it is not a simple
replication of the consolidation phase, although overlaps between the
underlying mechanisms of these two phases exist. Studies in rodents
have demonstrated that memory may be disrupted via inhibition of new
gene expression or protein synthesis in the hippocampus immediately
after retrieval of a consolidated memory, whereas the same
manipulations into the hippocampus long after retrieval had no effect
on memory. This indicates that these manipulations are effective only
when given soon after reactivation but not when given after a delay.
In addition, consolidated memories that are not retrieved, and
therefore do not become labile, cannot be disrupted by the inhibition
of gene expression or protein synthesis. Hence, if a consolidated
memory could turn back to its labile state and be disrupted by
external interventions, some authors argue that there might be the
chance that the disruption of the original traumatic memories could be
an efficacious approach to treat PTSD patients."
Memory extinction was first commented upon by Pavlov in 1927.
Today...
"Extinction training is not the same as forgetting, but it is
considered as new learning because it leaves the CS as having two
meanings: 'fear' if coupled with the US or 'safe' if it is not coupled
with US. When memory is extinguished, it means that the 'safe'
prevails over the 'fear' meaning. Many studies have provided evidence
demonstrating that extinction consists of a new learning process.
Specifically, without extinction training, the fear response lasts
months or even years, and extinguished fear responses return naturally
with the passage of time (spontaneous recovery), after using a
different context compared with the one in which extinction training
took place (renewal), or as a consequence of the re-exposure to the US
(reinstatement)"
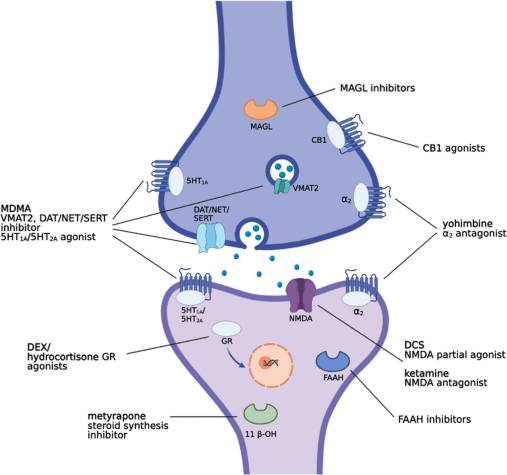
The authors'
"Literature search was conducted in March, 2021 by comprehensive
searches in two online databases (PubMed and Scopus). The keywords
used for the search of animal studies were: D-cycloserine, yohimbine,
methylene blue, glucocorticoids or corticosterone or methyrapone,
cannabinoids or endocannabinoids, MDMA or 3-4,
methylenedioxymethamphetamine, ketamine, psychedelics and/in post
traumatic stress disorder or posttraumatic stress disorder or
posttraumatic stress disorder or fear memory or memory extinction or
memory reconsolidation. Results were limited to rodents studies. The
keywords used for the search of human studies were: D-cycloserine,
yohimbine, methylene blue, glucocorticoids or cortisol, cannabinoids
or endocannabinoids, MDMA or 3-4, methylenedioxymethamphetamine,
ketamine, psychedelics and/in post traumatic stress disorder or
posttraumatic stress disorder or post-traumatic stress disorder. For
both the 1st (i.e., titles and abstracts) and the 2nd (i.e., full-text
articles) screening phases, the following exclusion criteria were
used: (1) papers written in a language other than English; (2)
non-original researches (e.g., reviews, commentaries, editorials, book
chapters); (3) no full-text articles (e.g., meeting abstracts); (4)
studies in vitro; (5) studies in non-human animals other than rodents;
(6) drugs administration not associated with an animal model of PTSD
or psychological intervention in humans; (7) absence of control groups
in the studies."
As for cannabinoids:
"The eCBs AEA and 2-AG are mainly degraded by two enzymes fatty-acid
amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL),
respectively. Increasing number of animal studies suggests that eCB
signaling is critically involved in the extinction phase of fear
memory. Several reports have demonstrated that increased AEA
signaling, through inhibition of FAAH activity, induced an enhancement
of fear memory extinction. On the contrary, the administration of CB1
receptor antagonists impaired the extinction and reconsolidation of
fear memory. In accordance with these results, CB1 KO mice or mice
bearing the FAAH C385A polymorphism, which impairs the activity of
FAAH, showed reduced and enhanced fear memory extinction,
respectively, thus, supporting the hypothesis that strengthening AEA
signaling at CB1 receptors enhances fear extinction. In a PTSD-like
rat model, we have recently demonstrated that the FAAH inhibitor
URB597 or the direct cannabinoid receptor agonist WIN55,212-2 (Fig. 1)
enhanced fear extinction tested 16 days post-trauma, when administered
after three spaced extinction sessions, 7, 10 and 13 days after trauma
exposure. Both drugs showed beneficial effects, but only URB597 (0.1
mg/kg, i.p.) induced the best improvements by enhancing extinction
consolidation and restoring normal social behavior in traumatized rats
through indirect activation of CB1 receptors tested up to 36-37 days
post-trauma. Further, another recent study has confirmed these
findings in male rodents showing beneficial effects on fear extinction
of the FAAH inhibitor URB597. Contrarily to what has been found and
described above in males, increased AEA signaling at CB1 receptors
(Fig. 1), 60 min prior to extinction training in an auditory fear
conditioning paradigm, did not alter fear memory extinction in female
rats. However, when AEA signaling was increased at the transient
potential receptor of vanilloid type-1 channel (TRPV1R, another target
of AEA beyond CB1 receptors), it augmented freezing behavior at
extinction training and extinction retrieval. Increased 2- AG
signaling at CB1 receptors reduced freezing at extinction training in
females, without altering fear responses in males."
But in humans, they believe:
"Rigorous clinical studies regarding the efficacy of cannabinoids for
PTSD in conjunction with psychotherapy are still lacking."
As for psychedelics Marchetta et al say:
"Psylocibin, lysergic acid diethylamide (LSD) and dimethyltryptamine
share a common mechanism of agonism at serotonergic 5-HT2A receptor.
Although few studies have indicated possible use of these psychedelics
for depression and obsessive-compulsive disorder, no other study has
yet been reported regarding a potential treatment for PTSD. However,
the effects on empathy, mindfulness related capacities, avoidance,
acceptance and connectedness, long-term openness, and emotional
break-through experiences along with preclinical studies,
demonstrating an enhancing effect on fear memory extinction, should
determine a renewed interest on these compounds."
https://pmc.ncbi.nlm.nih.gov/articles/PMC10207915/
[2490]
But were those clinical studies really "still lacking" in 2023?
The Prosecution should have been fully aware, by the early 2000s, of
the implications of taking away phytocannabinoids it said had no
medical utility:
"Seminal research in the early 2000s revealed that, in vitro, the
glucocorticoid-based inhibition of paraventricular nucleus neurons and
the subsequent inhibition of corticotropin-releasing hormone synthesis
depended on retrograde endocannabinoid signalling. Research in the
early 2000s also found that corticotropin-releasing hormone was found
to be highly co-expressed with CB1 receptors in the forebrain. It was,
therefore, suspected that endocannabinoids could be involved in the
regulation of the hypothalamic-pituitary-adrenal (HPA) axis response
to stress. Subsequent research confirmed this hypothesis, as it was
discovered that a reduction in AEA in the basolateral amygdala is
required for HPA axis activation, suggesting that AEA tonically gates
the HPA response. Research further explored the mechanisms involved in
HPA gating by AEA and found that CRH-mediated increases in FAAH
expression is responsible for the decrease in amygdala AEA prior to
HPA onset.
"While AEA is believed to tonically gate the HPA stress response, 2-AG
is believed to be involved in the termination of the response through
the facilitation of glucocorticoid-mediated negative feedback.
Research that supports this hypothesis has shown that 2-AG levels
increase in the hippocampus 30 to 60 min following stress, while the
inhibition of CB1 in the frontal areas of the brain prevents negative
feedback of the HPA axis. Likewise, the inhibition of MAGL, but not
FAAH, maintains increased corticosterone expression after stress
onset. Given the evidence described above, it has therefore been
widely hypothesised that endocannabinoids are involved in both the
initiation and termination of the HPA axis response to stress." [5126]
The Court is invited to evaluate the validity of the statement that
"no other study has yet been reported regarding a potential treatment
for PTSD" [2490] with psychedelics. No one has scientifically proved that I will
cheer up if I have a cup of tea. But that doesn't mean that I won't.
Then, in September 2023, Cell Reports published a study. Here are
some key facts, as reported by neurosciencenews.com:
"Summary: A recent study illuminates the brain's unique response to
stress: releasing its own cannabinoid molecules akin to THC from
cannabis plants.
"Centered in the amygdala, these molecules counteract stress alarms
originating from the hippocampus, an integral memory and emotion
region. This hints at the body's intrinsic mechanism for stress
management.
"Disruption in this system might escalate risks for stress-induced
psychiatric conditions.
"Key Facts:
"The amygdala, an emotional brain center, releases endogenous
cannabinoids to mitigate stress, interacting with the same receptors
as THC.
"When the target cannabinoid receptor was removed in mice, there was
a noticeable decline in their ability to handle stress and a
diminished inclination towards rewarding experiences.
"This study further supports the endocannabinoid system's role as a
potential drug-development candidate for stress-related
disorders."
https://neurosciencenews.com/endocannabinoids-stress-23913/
[3011]
According to the paper:
"BLA activity promotes eCB release at vHPC-BLA synapses
"Active stress coping recruits vHPC-BLA eCB signaling
"vHPC-BLA CB1 receptor deletion exacerbates stress-induced
avoidance/anhedonia
"The endocannabinoid (eCB) system is a key modulator of glutamate
release within limbic neurocircuitry and thus heavily modulates stress
responsivity and adaptation. The ventral hippocampus
(vHPC)-basolateral amygdala (BLA) circuit has been implicated in the
expression of negative affective states following stress exposure and
is modulated by retrograde eCB signaling. However, the mechanisms
governing eCB release and the causal relationship between vHPC-BLA eCB
signaling and stress-induced behavioral adaptations are not known.
Here, we utilized in vivo optogenetic- and biosensor-based approaches
to determine the temporal dynamics of activity-dependent and
stress-induced eCB release at vHPC-BLA synapses. Furthermore, we
demonstrate that genetic deletion of cannabinoid type-1 receptors
selectively at vHPC-BLA synapses decreases active stress coping and
exacerbates stress-induced avoidance and anhedonia phenotypes. These
data establish the in vivo determinants of eCB release at limbic
synapses and demonstrate that eCB signaling within vHPC-BLA circuitry
serves to counteract adverse behavioral consequences of stress."
Vanderbilt University, Tennessee, authors Kondev et al (2023) say
where this gets us to:
"A key question arises from our previous data demonstrating
relatively enhanced DSE at vHPC-BLA synapses in stress-resilient mice
and from our current data revealing 2-AG release at vHPC-BLA synapses
in response to stress exposure: is 2-AG-CB1R signaling at vHPC-BLA
synapses causally linked to stress-induced behavioral reactivity and
adaptation? Our data address this question in two separate, but
related, ways. First, we found that 2-AG release occurred during bouts
of active stress coping that were preceded by increases in BLA neuron
activity. Combined with previous data suggesting that BLA activity (or
activity of a subset of BLA output neurons) is associated with active
behavioral responses to stress, this suggests a possible scenario
whereby increases in BLA activity drive 2-AG release to dampen vHPC
afferent excitation via activation of presynaptic CB1R. That deletion
of CB1R from vHPC-BLA terminals reduced active stress coping in a
variety of assays (restraint stress, TST, FST) suggests that vHPC
inputs to the BLA promote passive stress coping. This hypothesis is
consistent with data demonstrating that vHPC-BLA circuits are critical
for driving freezing responses to contextual threat. How vHPC inputs
to the BLA selectively drive 'passive' output circuits or inhibit
'active' output circuits from the BLA is not known but represents an
important open question. Second, we found that deletion of CB1R from
vHPC-BLA synapses promotes anhedonia and avoidance selectively after
stress exposure and, more robustly, after repeated stress exposure.
These data suggest that 2-AG released within the synapse during stress
exposure plays an important role in buffering against subsequent
adverse behavioral consequences of stress, consistent with previous
studies. Taken together, these data indicate that 2-AG-CB1 signaling
at vHPC-BLA synapses plays an important role in regulating behavioral
responses during and after stress exposure and provides further
support for the contention that 2-AG signaling within defined limbic
circuits is a critical mechanism buffering against the adverse effects
of stress exposure. A key question arises from our previous data
demonstrating relatively enhanced DSE at vHPC-BLA synapses in
stress-resilient mice and from our current data revealing 2-AG release
at vHPC-BLA synapses in response to stress exposure: is 2-AG-CB1R
signaling at vHPC-BLA synapses causally linked to stress-induced
behavioral reactivity and adaptation? Our data address this question
in two separate, but related, ways. First, we found that 2-AG release
occurred during bouts of active stress coping that were preceded by
increases in BLA neuron activity. Combined with previous data
suggesting that BLA activity (or activity of a subset of BLA output
neurons) is associated with active behavioral responses to stress,
this suggests a possible scenario whereby increases in BLA activity
drive 2-AG release to dampen vHPC afferent excitation via activation
of presynaptic CB1R. That deletion of CB1R from vHPC-BLA terminals
reduced active stress coping in a variety of assays (restraint stress,
TST, FST) suggests that vHPC inputs to the BLA promote passive stress
coping. This hypothesis is consistent with data demonstrating that
vHPC-BLA circuits are critical for driving freezing responses to
contextual threat. How vHPC inputs to the BLA selectively drive
'passive' output circuits or inhibit 'active' output circuits from the
BLA is not known but represents an important open question. Second, we
found that deletion of CB1R from vHPC-BLA synapses promotes anhedonia
and avoidance selectively after stress exposure and, more robustly,
after repeated stress exposure. These data suggest that 2-AG released
within the synapse during stress exposure plays an important role in
buffering against subsequent adverse behavioral consequences of
stress, consistent with previous studies.
"Taken together, these data indicate that 2-AG-CB1 signaling at
vHPC-BLA synapses plays an important role in regulating behavioral
responses during and after stress exposure and provides further
support for the contention that 2-AG signaling within defined limbic
circuits is a critical mechanism buffering against the adverse effects
of stress exposure."
https://www.cell.com/cell-reports/fulltext/S2211-1247(23)01038-0
[3012]
In their 2022 paper "Neuroinflammation in Post-Traumatic Stress
Disorder" Dong-Hun Lee and colleagues at the Department of
Neurosurgery, College of Medicine, Soonchunhyang University, Cheonan
Hospital, Korea, pointed to pro-inflammatory cytokines as biomarkers
of PTSD:
"The Marine Resiliency Study reported that preexisting concentrations
of C-reactive protein (CRP) were directly correlated with the
occurrence and severity of PTSD, three months after a seven-month
military deployment, and elevated levels of interleukin (IL)-6, IL-8,
and transforming growth factor β (TGF-β) during
hospitalization predicted the development of PTSD one month later.
These data suggest that immune dysregulation predisposes individuals
to PTSD. The elevation of inflammatory cytokines in PTSD has clinical
significance, as chronic inflammation can adversely affect
cardiovascular and physical health. In addition, individuals with PTSD
are significantly more likely to suffer autoimmune disorders compared
with those with other psychiatric conditions. Inflammation-related
mediators, such as IL-1, IL-6, and tumor necrosis factor alpha
(TNF-α), may pass through the blood-brain barrier, and the
overproduction of pro-inflammatory cytokines can activate brain
microglia. A few small studies have examined the cerebrospinal fluid
levels of cytokines in PTSD but have yielded conflicting
results."
We like the large studies.
"One of the largest studies exclusively of men exposed to combat
trauma reported a significantly elevated pro-inflammatory composite,
comprising IL-1β, IL-6, TNF-α, IFN-γ, and CRP levels
in those with PTSD compared to those without PTSD. The predominant
cytokines were different for each group, and differences were observed
in TNF-α, IFN-γ, and IL-6 values. The pro-inflammatory
cytokine levels remained significantly higher in individuals with PTSD
after controlling for early-life trauma, major depressive disorder and
its severity, body mass index, ethnicity, education, asthma and/or
allergies, time since combat, potentially confounding inflammatory
illnesses, and medications. Immune activation of PTSD was also found
to be significantly high in male groups who experienced combat
trauma.
"Conversely, several other studies have reported no significant
difference in inflammatory marker levels between patients with PTSD
and healthy controls."
Lee et al discuss the role of DAMP, HPA axis, serotonin and
tryptophan-kynurenine pathways:
"Damage-associated molecular patterns (DAMPs) are host-derived
non-bacterial immune response molecules released due to PTSD-related
stress. These molecules act as danger signals when cells are damaged.
When DAMPs are released from cells, pattern recognition receptors
(PRRs) are expressed in astrocytes and microglia to promote
neuroinflammatory reactions. The PRR is a protein expressed by innate
immune system cells such as dendritic cells, macrophages, monocytes,
and neutrophils, and is a receptor that recognizes DAMPs and plays an
important role in innate immunity. One of the PRRs, the Toll-like
receptor (TLR), is expressed in various cells of the CNS, including
neurons, microglia, and astrocytes. These PRRs are activated by
detecting endogenous molecules such as DAMPs, and the persistent
neuroinflammatory action of glial cells can lead to
neurodegeneration."
and
"IL-1 receptors (IL-1Rs) are produced by microglia, astrocytes, and
brain oligodendrocytes, and have been shown to be highly concentrated
in the pituitary and meninges. IL-1R mRNA is expressed in the
capillaries and glial cells throughout the hippocampus, choroid
plexus, cerebellum, and brain, and the IL-1 type 1 receptor is
expressed in humans' hypothalamus. The secreted inflammatory cytokines
are received in these glial cells and contribute to neuroinflammation.
Therefore, the impairment of the HPA axis due to PTSD can lead to
neuroinflammation."
and
"Tryptophan is an essential amino acid used in protein biosynthesis
and is mainly metabolized with kynurenine. Kynurenine is a metabolite
of tryptophan that performs a variety of biological functions,
including vasodilation and immune response during inflammation. This
kynurenine pathway is modified in several diseases, including mental
disorders such as schizophrenia and depressive disorders.
Pro-inflammatory cytokines can enhance the activity of indolamine
2,3-dioxigenase (IDO), the first and rate-limiting enzyme in the
tryptophan degradation pathway. Elevated levels of pro-inflammatory
cytokines increase IDO enzyme activity. IDO is involved in tryptophan
metabolism, which upregulates the production of kynurenine. Kynurenine
is converted into several metabolites, including quinolinic and
kynurenic acids, and it is involved in the activity and inhibition of
NMDA (N-methyl-d-aspartate), with elevated pro-inflammatory cytokines
producing relatively larger amounts of quinolinic acid (Figure 4).
This increase in quinoline acid has a potent neurotoxic effect, which
can activate microglia and macrophages, increase reactive oxygen and
reactive nitrogen species due to lipid peroxidation, and interfere
with nerve function or cause apoptosis. Some studies have also shown
that quinoline acid may also be linked to mental disorders, although
the mechanism is still unclear. However, quinoline acid has been found
in the postmortem brain of depressed patients."
The authors briefly mention cannabis, and claim rather
ignorantly
"...very few studies have reported the potential usefulness of
cannabis in the treatment of PTSD."
In Korea, perhaps. There are only 13.9 million Google results for
"cannabis ptsd korea". However these authors suggest a variety of drug
treatments including NSAIDs, steroids, angiotensin-converting enzyme
inhibitors and angiotensin receptor blockers, and invasive treatments
like deep brain stimulation (DBS).
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9138406/
[4076]
In 2007 Miller and Wrosch found that carrying on with unattainable
goals appears to affect C-reactive protein.
"The notion that persistence is essential for success and happiness
is deeply embedded in popular and scientific writings. However, when
people are faced with situations in which they cannot realize a key
life goal, the most adaptive response for mental and physical health
may be to disengage from that goal. This project followed 90
adolescents over the course of 1 year. Capacities for managing
unattainable goals were assessed at baseline, and concentrations of
the inflammatory molecule C-reactive protein (CRP) were quantified at
that time, as well as 6 and 12 months later. To the extent that
subjects had difficulties disengaging from unattainable goals, they
displayed increasing concentrations of CRP over the follow-up. This
association was independent of potential confounds, including
adiposity, smoking, and depression. Because excessive inflammation
contributes to a variety of adverse medical outcomes, these findings
suggest that in some contexts, persistence may actually undermine
well-being and good health."
https://pubmed.ncbi.nlm.nih.gov/17760771/
[4698]
We underestimate the effect of trauma on third parties, as shown by
Song et al in "Firearm Injuries In Children And Adolescents: Health
And Economic Consequences Among Survivors And Family Members"
(2023):
"More US children and adolescents today die from firearms than any
other cause, and many more sustain firearm injuries and survive. The
clinical and economic impact of these firearm injuries on survivors
and family members remains poorly understood. Using 2007-21 commercial
health insurance claims data, we studied 2,052 child and adolescent
survivors compared to 9,983 matched controls who did not incur firearm
injuries, along with 6,209 family members of survivors compared to
29,877 matched controls, and 265 family members of decedents compared
to 1,263 matched controls. Through one year after firearm injury,
child and adolescent survivors experienced a 117 percent increase in
pain disorders, a 68 percent increase in psychiatric disorders, and a
144 percent increase in substance use disorders relative to the
controls. Survivors' health care spending increased by an average of
$34,884—a 17.1-fold increase—with 95 percent paid by
insurers or employers. Parents of survivors experienced a 30-31
percent increase in psychiatric disorders, with 75 percent more mental
health visits by mothers, and 5-14 percent reductions in mothers' and
siblings' routine medical care. Family members of decedents
experienced substantially larger 2.3- to 5.3-fold increases in
psychiatric disorders, with at least 15.3-fold more mental health
visits among parents. Firearm injuries in youth have notable health
implications for the whole family, along with large effects on
societal spending."
https://www.healthaffairs.org/doi/abs/10.1377/hlthaff.2023.00587
[4085]
Clearly, individuals vary in their susceptibility to PTSD for a
variety of reasons. Unusual long-term background stressors and acute
trauma have conspired against the Defendant's well-being fully
justifying continued use of both cannabis and psychedelics with no
need for therapy. Indeed in one unpopular paper it was shown that
therapists add no value to psychedelics: "no significant association
was found between the number of therapy hours and outcome in either
the short-term (b = −0.05, p = .327) or long-term."
https://www.sciencedirect.com/science/article/pii/S0163834325001562
[5294]
In the view of the Defendant and most illicit users, therapy is a
two-edged sword, as likely to undermine the confidence of the subject
as rebuild it.
Exploitation is a real probability, best averted not by having no
unsupervised psychedelicism, but by having no therapist.
Psychiatrists reinforce the sick role.
150 years ago the concept did not really exist.
Psychedelic therapy could make someone else legally and morally liable
for a person's mistakes.
Therapists are more expensive than real friends.
Therapists may try to do too many things at once, not all of them for
the "patient".
As in the wider field of psychiatry, psychedelic therapy is an attempt
at labelling all users as mentally ill in one way or another.
And with those endless categories and theories, and mental confusion
about "drugs", even with mushrooms welcomed back into the
pharmaceutical fold, psychiatrists can start out meaning well...but
they face difficulties being part of the set and setting of a
therapeutic journey, with psychedelics or without.
9 to 40 million years after the first biosynthesis of psilocybin [3252] individual vulnerability to PTSD was addressed by Suprani et al
[2025]:
"While the symptom burden can be substantial-particularly in high-risk
populations exposed to cumulative or prolonged trauma-the lifetime
prevalence of full-syndrome PTSD in the general population remains
lower; for instance, it is estimated at 6.1% in the United States.
Epidemiological evidence has consistently shown that only a minority
of trauma exposed individuals go on to develop PTSD. The
neurobiological mechanisms underpinning the individual variation in
vulnerability to PTSD development following trauma are still unclear.
Different biological systems have been investigated, such as those
involved in neuroplasticity and immunoregulation. More recently, the
endocannabinoid (eCB) system has received attention in the PTSD
research field, given its modulatory effect on memory consolidation,
retrieval and extinction. There is a large amount of data in animal
models supporting the eCB system role in regulating fear and stress
responses. The fatty acid-derived signaling molecule Anandamide (AEA),
the first discovered endocannabinoid, was shown to facilitate fear
extinction in mice by activating the Cannabinoid receptor 1 [CB1].
When research on the eCB system in fear responses was extended to
humans, consistent results were found, mostly on healthy subjects.
Three different human studies replicated the association between
enhanced fear extinction and a hypofunctional genetic variation in the
fatty acid amide hydrolase (FAAH), an enzyme that catabolizes AEA.
Positive results in genetics research were also published by Heitland
et al., who found that A/A carriers of rs2180619 (a polymorphism
located in the promoter region of CB1), exhibited impaired fear
extinction. In line with preclinical and healthy volunteers'
literature, individuals with PTSD were shown to have a reduction in
circulating endocannabinoids levels.
"A recent study identified the amygdala-projecting medial prefrontal
cortex (mPFC) neurons as a potential neural basis for the effect of
the eCB system on fear extinction. The amygdala is a primary hub in
the brain that regulates the processing of aversive memories, by
integrating information from different cortical brain regions. Among
these regions, the mPFC was shown to exert top-down control of
negative emotions over the amygdala in mice. Findings in patients with
PTSD suggest that the interaction between the mPFC and the amygdala
becomes dysfunctional, resulting in the heightened responsivity of the
latter. Several recent studies with different designs support this
hypothesis. For instance, neurosurgical patients with ventral mPFC
lesions exhibited potentiated amygdala responses to aversive images
compared to neurologically healthy controls. Consistently, Feng et
al., showed that the resting-state functional connectivity between the
ventral mPFC and the amygdala is positively correlated with fear
extinction after fear reminder in healthy subjects. Overall, numerous
lines of evidence point toward a potential role of the eCB system in
PTSD vulnerability following trauma, possibly via altered top-down
control on the amygdala."

https://www.frontiersin.org/journals/psychiatry/articles/10.3389/fpsyt.2025.1584583/full
[5295]
In "The Effectiveness and Adverse Events of Cannabidiol and
Tetrahydrocannabinol Used in the Treatment of Anxiety Disorders in a
PTSD Subpopulation" (2023) from Stack et al of the University of
Sydney's School of Pharmacology and Applied Cannabis Research in
Sydney, NSW, Australia:
"The median doses taken were 50.0 mg/day for CBD and 4.4 mg/day for
THC. The total participant sample reported significantly improved
anxiety, depression, fatigue, and ability to take part in social roles
and activities. Those who were diagnosed with PTSD (n = 57) reported
significantly improved anxiety, depression, fatigue, and social
abilities. The most common AEs reported across the whole participant
cohort were dry mouth (32.6%), somnolence (31.3%), and fatigue
(18.5%), but incidence varied with different cannabis formulations.
The inclusion of THC in a formulation was significantly associated
with experiencing gastrointestinal AEs; specifically dry mouth and
nausea. Conclusions: Formulations of cannabis significantly improved
anxiety, depression, fatigue, and the ability to participate in social
activities in participants with anxiety disorders. The AEs experienced
by participants are consistent with those in other studies."
And of special note:
"Participants who took a THC-dominant formulation reported a
significant decrease in their anxiety levels. Those same participants
also represented the highest proportion of participants that were
classified as having clinical improvement (61.1%, n = 11), compared
with participants who were prescribed other formulation types. This
was unexpected as the median THC dose for this participant group was
33.8 mg/day, and it has been suggested that doses higher than 30
mg/day could be anxiogenic.14 A study reported that doses of CBD
ranging from 15 to 60 mg/day could offset the anxiogenic properties of
THC, which is reflected in our data; however, with lower doses of CBD
(median = 6.0 mg/day CBD)."
https://journals.sagepub.com/doi/pdf/10.1177/87551225231180796
[4557]
epidemic declared March 12 then again October 19 2020
102 C/C polymorphism and suicide
Several lines of evidence indicate that abnormalities in the
functioning of the central serotonergic system are involved in the
pathogenesis of depressive illness and suicidal behavior.
https://pubmed.ncbi.nlm.nih.gov/19224112/
[298]
https://pubmed.ncbi.nlm.nih.gov/29526601/
[299]
https://pubmed.ncbi.nlm.nih.gov/11727894/
[300]
Research does not seek to eliminate environmental influences in
favour of genetic disposition in depression, suicidal ideation and
completed suicide. But there are only two hypotheses for the high
incidence in Slovenia, both of which may be right, namely that some
are genetically predisposed to snap when stress is prolonged or
reaches a certain level, or that living conditions for some, according
to the rules constructed by their own society, are relentlessly
sub-optimal.
Slovenia is an ideal candidate for psychedelic therapy. Slovenia
ruminates endlessly about the past. Millions of tonnes of CO2 are
expended generating hot air about long-dead regimes and ideologies,
because they have affected people in this area. But arguing about what
Tito wrote about Stalin in 1940 won't bring your dead relatives back
or make you happy and strong today. To ignore the past and bury the
hatchet seems like a cop-out. But the past is getting in the way of
the present, and the future.
"In 1976, a man known as Ka-Tzetnik 135633 (original name: Yehiel
De-Nur) underwent LSD-assisted psychotherapy. At the time, he was
under the supervision of Prof. Jan Bastiaans at the State University
of Leiden in the Netherlands. Bastiaans was the psychiatrist who had
first identified concentration camp syndrome, also known as survivor
syndrome. This syndrome is a form of what we would now call
post-traumatic stress disorder (PTSD). Its characteristics include
social withdrawal, sleep disturbance, anxiety, and depression.
Bastiaans had also successfully treated this syndrome with
LSD-assisted psychotherapy. Ka-Tzetnik 135633 was a survivor of
Auschwitz, where he was a prisoner for two years. This trauma was
followed by three decades of torment, in which he relived the horror
of those two years at night. Through LSD-assisted psychotherapy,
however, Ka-Tzetnik 135633 found healing.
"Almost a half century after Bastiaans' successful trials, the
regulation of psychedelics for treating PTSD is finally underway.
Think of the Holocaust survivors who might have found healing through
these medicines, but couldn't because of a politically-motivated ban
on psychedelics. This realization brings home the horrific impact of
the failed war on drugs.'"
https://realitysandwich.com/psychedelics-and-judaism/
[1092]
Psychedelics break the rumination cycle by disrupting - in a good way
- brain activities which perpetuate habitual thinking and behaviour.
The most abundant 5-HT receptors expressed in the medial prefrontal
cortex (mPFC) are 5-HT1AR and 5-HT2AR. The 5-HT2AR pathway is enhanced
by 5-HT2AR-agonist psychedelics.
https://pubmed.ncbi.nlm.nih.gov/28858536/
[301]
In mice, given a single dose of psilocybin or LSD, and using the
social reward conditioned place preference (sCPP) assay Nardou et al
have made an important step forward in understanding the mechanism by
which psychedelics can override cultural log-jams retarding progress
and societal happiness, such as we see in Slovenia. "Psychedelics
reopen the social reward learning critical period" (2023)
finds:
"Psychedelics are a broad class of drugs defined by their ability to
induce an altered state of consciousness. These drugs have been used
for millennia in both spiritual and medicinal contexts, and a number
of recent clinical successes have spurred a renewed interest in
developing psychedelic therapies. Nevertheless, a unifying mechanism
that can account for these shared phenomenological and therapeutic
properties remains unknown. Here we demonstrate in mice that the
ability to reopen the social reward learning critical period is a
shared property across psychedelic drugs. Notably, the time course of
critical period reopening is proportional to the duration of acute
subjective effects reported in humans. Furthermore, the ability to
reinstate social reward learning in adulthood is paralleled by
metaplastic restoration of oxytocin-mediated long-term depression in
the nucleus accumbens. Finally, identification of differentially
expressed genes in the 'open state' versus the 'closed state' provides
evidence that reorganization of the extracellular matrix is a common
downstream mechanism underlying psychedelic drug-mediated critical
period reopening. Together these results have important implications
for the implementation of psychedelics in clinical practice, as well
as the design of novel compounds for the treatment of neuropsychiatric
disease."
https://www.nature.com/articles/s41586-023-06204-3
[2825]
But we do not need to rely on mice when there are real-life mass
traumas going on. A special opportunity came along on 7 October 2023
in the Hamas attack on Supernova. "Trauma Under Psychedelics: MDMA
Shows Protective Effects During the Peritraumatic Period" say Netzer
et al (2024):
"Traumatic events (TEs) play a causal role in the etiology of
psychopathologies such as depression and posttraumatic stress disorder
(PTSD). Recent research has highlighted the therapeutic potential of
psychoactive substances and especially
3,4-methylenedioxymethamphetamine (MDMA), in alleviating trauma
symptoms in chronic patients. However, little is known regarding the
consequences of trauma that is acutely experienced under the influence
of psychoactive substances. Here we investigated the acute experiences
and peritraumatic processing of 657 survivors from the high-casualty
terror attack at the Supernova music festival in Israel on October
7th, 2023. Data were collected four to twelve weeks following the TE.
Approximately two-thirds of survivors were under the influence of
psychoactive substances at the time of the TE, offering a tragic and
unique natural experiment on the impact of psychoactive compounds on
TE processing. Our findings reveal that individuals who experienced
the trauma while under the influence of MDMA demonstrated
significantly improved intermediate outcomes compared to those who
were under the influence of other substances or no substances at all.
Specifically, the MDMA group reported increased feelings of social
support, more social interactions and enhanced quality of sleep during
the peritraumatic period, yielding reduced levels of mental distress
and reduced PTSD symptom severity. These novel findings suggest that
the influence of MDMA during the TE may carry protective effects into
the peritraumatic period, possibly mediated through the known effects
of MDMA in reducing negative emotions and elevating prosociality.
These protective effects in turn may mitigate the development of early
psychopathology-related symptoms. Current preliminary results
underscore the need for further understanding of the cognitive and
physiological processes by which psychedelic substances intersect with
trauma recovery processes."
https://www.biorxiv.org/content/10.1101/2024.03.28.587237v1
[3174]
Another alternative to mice is "Human pluripotent stem cells as a
translational toolkit in psychedelic research in vitro". As Salerno
and Rehan explain:
"PSCs can differentiate into various brain cell types, mirroring
endogenous expression patterns and cell identities to recreate disease
phenotypes. Brain organoids derived from PSCs resemble cell diversity
and patterning, while region-specific organoids simulate circuit-level
phenotypes. PSC-based models hold significant promise to illuminate
the cellular and molecular substrates of psychedelic-induced
phenotypic recovery in neuropsychiatric disorders."
and
"The identities of the signaling pathways responsible for
psychedelics' hallucinogenic and therapeutic properties and whether
these are distinct or overlapping pathways are some of the questions
still under debate.
"The Gq or β-arrestin-2 recruitment paradigm represents a
glimpse into the extensive network of pathways regulated by 5-HT2AR
activation via psychedelics. For example, distinct psychedelics
activate phospholipase A2 and phospholipase D via ADP-ribosylation
factor 1. The activation of 5-HT2AR, triggered by various agonists
including DOI and LSD, can lead to the heterodimerization of the
receptor with metabotropic glutamate receptor 2 and dopamine D2
receptor, both of which are Gi-coupled receptors. This transactivation
blocks the cAMP's synthesis by adenylate cyclase and prompts
heterotrimeric Gi/o proteins to trigger Src-mediated downstream
events.
"A burst of glutamate also follows psychedelic administration, mainly
in the cortical layer V of the neocortex, which is a 5-HT2AR-enriched
area.8,23 High glutamate levels activate the
α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor
(AMPAR), boosting the secretion of brain-derived neurotrophic factor
(BDNF), which in turn signals through the tyrosine receptor kinase B
(TrkB) and mammalian target of rapamycin (mTOR) pathways, sustaining
both the AMPAR activation and BDNF secretion in a positive feedback
loop of neural plasticity.
"Furthermore, the hallucinogenic properties of different psychedelics
are also proposed to be influenced by other 5-HTRs, and not all
5-HT2AR agonists have hallucinogenic properties. For instance, human
studies also provide evidence supporting the involvement of the
5-HT1AR in the effects of psilocybin. Competition binding studies on
rodent brains and primary cells revealed a lack of pronounced
selectivity of psilocin for 5-HT2AR over 5-HT1AR. Nevertheless, the
role of the 5-HT2AR remains the subject of more extensive study and
investigation. Ketanserin, a selective antagonist of the 5-HT2Rs,
effectively eliminates head-twitch behavioral responses in mice;
however, it does not attenuate psilocybin-induced structural
modifications in the prefrontal cortex. As a result, uncertainties
remain about whether and how the neuroplastic effects on rescuing
disease phenotypes relate to behavioral responses, especially in
humans. Moreover, human studies investigating the molecular mechanisms
of psychedelic-induced plasticity rely on global parameters such as
peripheral BDNF levels, which may not correlate with the molecular
mechanisms occurring in brain cells. For in-depth cellular and
molecular insights into the role of 5-HT2AR, in vitro investigations
are required."
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC11019282/ [3173]
Using PSCs Schmidt et al (2024) of the University of Heidelberg
showed that psilocin causes 5-HT2A receptor internalization and
redistribution in human cortical neurons, induces BDNF expression and
downstream signaling in human neurons in a dose-dependent manner,
induces gene expression changes associated with axonal and synaptic
plasticity, increases dendritic complexity, and increases synaptic
strength and synaptogenesis.
https://www.researchgate.net/publication/381253376_Psilocin_fosters_neuroplasticity_in_iPSC-derived_human_cortical_neurons
[3175]
Administration of psychedelics to a relatively small number of people
could bring the paralysing stand-off over Slovenia's past to a quick
and elegant halt. The participants would see the damage they are doing
- to themselves and the country - by perpetuating hostilities against
groups and against individuals long dead. They would be able to more
accurately gauge the importance of the historical against the
challenges of the present and future. They would be less susceptible
to fearmongering and superstition. Their nightmares would be put into
perspective.
The Slovenians who stand to gain the most from ranting on about the
past for political purposes are the least likely to have taken them.
Among them you will find the most enthusiastic opponents of the
benefits of psychedelics, about which their ignorance resembles what
the average villager in Radenci knows of Tito in the 1940s, i.e. what
they have been told, not what they have experienced.
Unnecessary and counterproductive rigidity are legal and mental
constructs that can be ameliorated by mind-expanding drugs. They offer
a sideways view, and offer users the opportunity to invite unnecessary
and counterproductive rigidity to get lost. And this is why the
anti-drug religion, prohibition, doesn't like them.
Oblivion from alcohol, often associated with violence, is a legally
approved coping mechanism, but is solipsistic and does nothing to
solve the underlying malaises. Drugs other than alcohol, which can
give rise to creative solutions to life stresses or at least relief by
way of euphoria are legally disapproved, and the users accused of
escapism. A drug user is not concerned with his or her genetic
predisposition. They can't change that.
What Slovenia's warring factions need is not more press stories about
what the communists did, but consolation now. Now does not contain the
grudges of the past, nor the anxieties of the future. Now is not a
popular concept for political manipulators. "Now" suggests something
should be done, rather than talked about. "Now" cannot play on
people's resentments and fears. Dealing with "Now" may be financially
expensive compared to promises and grumbling.
Although it is not necessary to understand the mechanisms of the
human brain by which consolation takes place, and psychedelic people
have managed without this so far, it's good to know the scientists are
finally being allowed to fill in the details, and the activation of
5-HT1A receptors in the anterior cingulate cortex was sufficient to
reverse consolation and sociability deficits in Chinese mandarin
voles.
https://elifesciences.org/articles/67638
[302]
The Defence thinks it might work in Slovenians too.
----------------------------------------------------------------------------
The Englishman stands for the rights of everyone disadvantaged,
discriminated against, persecuted, and prosecuted on the false or
absent bases of prohibition, and also believes the victims of these
officially-sanctioned prejudices have been appallingly treated and
should be pardoned and compensated.
The Englishman requests the return of his CaPs and other rightful
property, for whose distraint Slovenia has proffered no credible
excuse or cause.
The Benedictions represent both empirical entities as well as beliefs.
Beliefs which the Defence evidence shows may be reasonably and
earnestly held about the positive benefits of CaPs at the population
level, in which the good overwhelmingly outweighs the bad. Below, the
latest version of this dynamic list.
THE BENEDICTIONS
REFERENCES
TIMELINE OF DRUG LAW v. SCIENCE














